

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50080092 (17beta)-17-hydroxyestr-4-en-3-one::17beta-hydroxy-19-nor-4-androsten-3-one::17beta-hydroxy-4-estren-3-one::17beta-hydroxyestr-4-en-3-one::19-Norandrostenolone::19-Nortestosterone::4-estren-17beta-ol-3-one::CHEMBL757::Nandrolone
SMILES: C[C@]12CC[C@H]3[C@@H](CCC4=CC(=O)CC[C@H]34)[C@@H]1CC[C@@H]2O
InChI Key: InChIKey=NPAGDVCDWIYMMC-IZPLOLCNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Corticosteroid-binding globulin (Homo sapiens) | BDBM50080092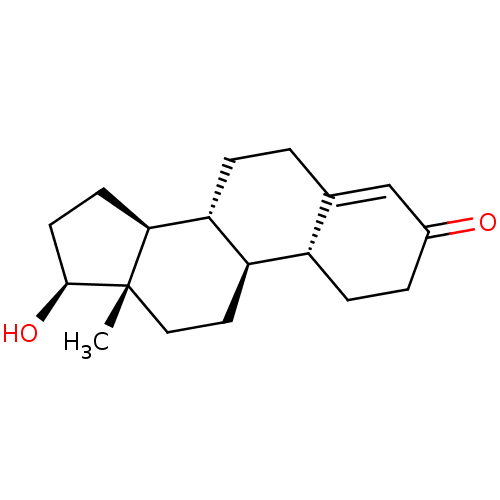 ((17beta)-17-hydroxyestr-4-en-3-one | 17beta-hydrox...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 724 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Zoki Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Binding affinity to human CBG receptor (corticosteroid-binding globulins) | J Med Chem 47: 2732-42 (2004) Article DOI: 10.1021/jm030364c BindingDB Entry DOI: 10.7270/Q2WM1H5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyruvate dehydrogenase kinase (Homo sapiens (Human)) | BDBM50080092 ((17beta)-17-hydroxyestr-4-en-3-one | 17beta-hydrox...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.20E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute for Biomedical Research Curated by ChEMBL | Assay Description In vitro inhibitory activity against pyruvate dehydrogenase kinase was determined | Bioorg Med Chem Lett 9: 2223-8 (1999) BindingDB Entry DOI: 10.7270/Q2RN3B1Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Testis-specific androgen-binding protein (Homo sapiens (Human)) | BDBM50080092 ((17beta)-17-hydroxyestr-4-en-3-one | 17beta-hydrox...) | PDB MMDB NCI pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 501 | n/a | n/a | n/a | n/a | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Displacement of [3H]5alpha dihydrotestosterone from human sex hormone binding globulin | J Med Chem 51: 2047-56 (2008) Article DOI: 10.1021/jm7011485 BindingDB Entry DOI: 10.7270/Q2RX9DC2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenergic Alpha (Homo sapiens (Human)) | BDBM50080092 ((17beta)-17-hydroxyestr-4-en-3-one | 17beta-hydrox...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.51E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University Curated by ChEMBL | Assay Description Inhibition of 4-(4-(dimethylamino)styryl)-N-methylpyridinium uptake at human OCT1 expressed in HEK293 cells by confocal microscopy | J Med Chem 51: 5932-42 (2008) Article DOI: 10.1021/jm8003152 BindingDB Entry DOI: 10.7270/Q23779MD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||