

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50080498 3-{(S)-3-[(7-Methoxy-naphthalene-2-sulfonyl)-thiophen-3-ylmethyl-amino]-2-oxo-pyrrolidin-1-ylmethyl}-benzamidine::CHEMBL119456
SMILES: COc1ccc2ccc(cc2c1)S(=O)(=O)N(Cc1ccsc1)[C@H]1CCN(Cc2cccc(c2)C(N)=N)C1=O
InChI Key: InChIKey=HNEAWVZXDQKQPL-SANMLTNESA-N
Data: 3 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor X (Homo sapiens (Human)) | BDBM50080498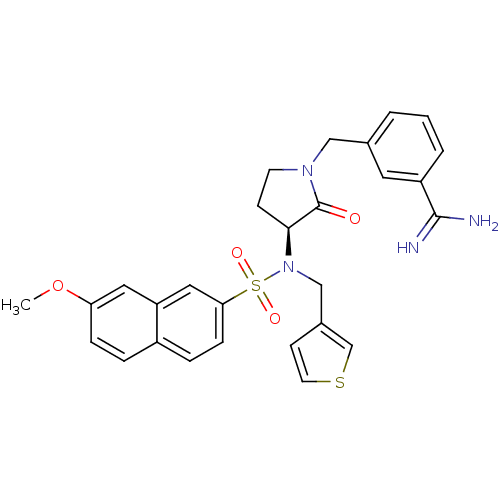 (3-{(S)-3-[(7-Methoxy-naphthalene-2-sulfonyl)-thiop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rh£ne-Poulenc Rorer Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of human Coagulation factor Xa | J Med Chem 42: 3557-71 (1999) Article DOI: 10.1021/jm990040h BindingDB Entry DOI: 10.7270/Q2M04640 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50080498 (3-{(S)-3-[(7-Methoxy-naphthalene-2-sulfonyl)-thiop...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rh£ne-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human thrombin | J Med Chem 42: 3557-71 (1999) Article DOI: 10.1021/jm990040h BindingDB Entry DOI: 10.7270/Q2M04640 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin II (Bos taurus) | BDBM50080498 (3-{(S)-3-[(7-Methoxy-naphthalene-2-sulfonyl)-thiop...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rh£ne-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibitory activity against bovine pancreatic trypsin | J Med Chem 42: 3557-71 (1999) Article DOI: 10.1021/jm990040h BindingDB Entry DOI: 10.7270/Q2M04640 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||