

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50083227 1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1,3-dihydro-benzoimidazol-2-one::CHEMBL146054
SMILES: CC(N1CCC(CC1)n1c2ccccc2[nH]c1=O)c1ccccc1Cl
InChI Key: InChIKey=ICPHYGRATJRGHH-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50083227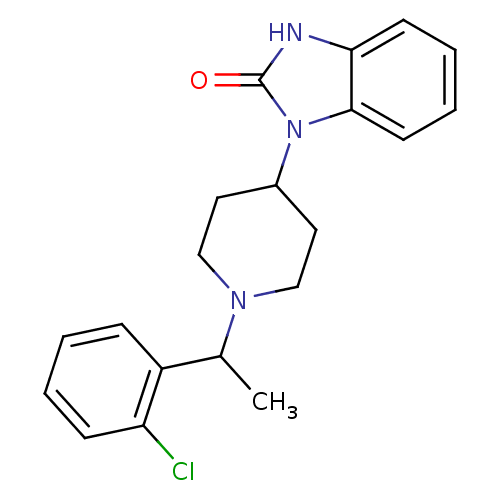 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity of the compound in CHO cells stably expressing cloned human Opioid receptor delta 1 by displacing radioligand [3H][D-Ala2,D-Leu5]enk... | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Antagonistic activity against nociceptin produced [35S]GTP-gamma-S, binding to Opioid receptor like 1 expressed in CHO cells | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Agonistic activity of the compound against nociceptin produced GTPgammaS binding to Opioid receptor like 1 expressed in CHO cells | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 199 | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Agonist activity at human mu opioid receptor expressed in human USOS-beta-arrestin-hMOR-PathHunter cells incubated for 90 mins by beta-arrestin-2 enz... | J Med Chem 61: 8895-8907 (2018) Article DOI: 10.1021/acs.jmedchem.8b01136 BindingDB Entry DOI: 10.7270/Q2862K2V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity in CHO cells stably expressing cloned human Opioid receptor mu 1 by displacing diprenorphine | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity of the compound in CHO cells stably expressing cloned human Opioid receptor kappa 1 by displacing radioligand [3H]-U-69,593 | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 31 | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Agonist activity at human mu opioid receptor expressed in CHO cell membranes assessed as stimulation of [35S]GTPgammaS binding incubated for 1 hr by ... | J Med Chem 61: 8895-8907 (2018) Article DOI: 10.1021/acs.jmedchem.8b01136 BindingDB Entry DOI: 10.7270/Q2862K2V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50083227 (1-{1-[1-(2-Chloro-phenyl)-ethyl]-piperidin-4-yl}-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity of the compound in CHO cells stably expressing cloned human Opioid receptor like 1 by displacing radioligand [125I]-Tyr14-nociceptin | J Med Chem 42: 5061-3 (2000) BindingDB Entry DOI: 10.7270/Q2MC8Z69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||