

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50089655 CHEMBL3576976
SMILES: Cc1ccc(cc1)S(=O)(=O)n1cc(C(=O)C(=O)N[C@@H](Cc2cnc[nH]2)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c2ccccc12
InChI Key:
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cyclooxygenase (Homo sapiens (Human)) | BDBM50089655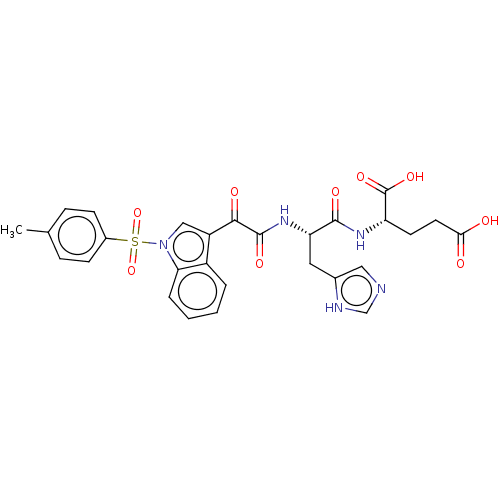 (CHEMBL3576976) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guru Nanak Dev University Curated by ChEMBL | Assay Description Competitive inhibition of human recombinant COX-2 by UV-Visible spectrophotometry | Eur J Med Chem 97: 104-23 (2015) Article DOI: 10.1016/j.ejmech.2015.04.044 BindingDB Entry DOI: 10.7270/Q2N87CJN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase (cyclooxygenase) (Ovis aries (Sheep)) | BDBM50089655 (CHEMBL3576976) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Guru Nanak Dev University Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 assessed as PGF2alpha formation using arachidonic acid as substrate pretreated with compound for 20 mins prior to substrate... | Eur J Med Chem 97: 104-23 (2015) Article DOI: 10.1016/j.ejmech.2015.04.044 BindingDB Entry DOI: 10.7270/Q2N87CJN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclooxygenase (Homo sapiens (Human)) | BDBM50089655 (CHEMBL3576976) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
Guru Nanak Dev University Curated by ChEMBL | Assay Description Inhibition of human recombinant COX-2 assessed as PGF2 alpha formation using arachidonic acid as substrate pretreated with compound for 20 mins prior... | Eur J Med Chem 97: 104-23 (2015) Article DOI: 10.1016/j.ejmech.2015.04.044 BindingDB Entry DOI: 10.7270/Q2N87CJN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||