

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50092152 2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-diaza-bicyclo[13.2.2]nonadeca-1(18),15(19),16-trien-13-ylamino)-4-hydroxy-pentanoic acid (2-hydroxy-indan-1-yl)-amide::CHEMBL331945
SMILES: CCC(C)[C@@H]1NC(=O)[C@H](Cc2ccc(OCCCCCNC1=O)cc2)NC[C@@H](O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H]1[C@H](O)Cc2ccccc12
InChI Key: InChIKey=ZXUOAWXMXWTRGE-ASZZIURCSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50092152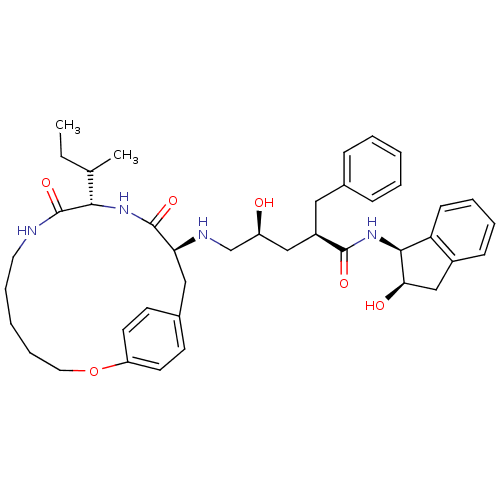 (2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-dia...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibitory constant against HIV-1 protease | J Med Chem 43: 3495-504 (2000) BindingDB Entry DOI: 10.7270/Q2KH0P1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50092152 (2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-dia...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of virion associated RT activity relative to untreated, infected control in MT2 cells infected with HIV-2 strain (ROD) | J Med Chem 43: 3495-504 (2000) BindingDB Entry DOI: 10.7270/Q2KH0P1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50092152 (2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-dia...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of virion associated RT activity relative to untreated, infected control in MT2 cells of HIV-1 237288 strain | J Med Chem 43: 3495-504 (2000) BindingDB Entry DOI: 10.7270/Q2KH0P1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50092152 (2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-dia...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of virion associated RT activity relative to untreated, infected control in cord blood mononuclear cells (CBMC) infected with HIV -1 TC354... | J Med Chem 43: 3495-504 (2000) BindingDB Entry DOI: 10.7270/Q2KH0P1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50092152 (2-Benzyl-5-(10-sec-butyl-9,12-dioxo-2-oxa-8,11-dia...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of virion associated RT activity relative to untreated, infected control in PBMC cells infected with HIV-1 237288 strain | J Med Chem 43: 3495-504 (2000) BindingDB Entry DOI: 10.7270/Q2KH0P1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||