

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CCc1ccc(OCc2ccccc2C(=O)Nc2ccc3nc(C)cc(N)c3c2)cc1
InChI Key: InChIKey=VTGBZWHPJFMTKS-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50094634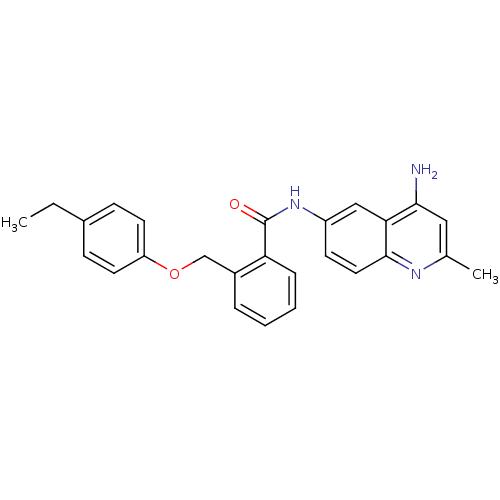 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Stimulation of [35S]GTP-gamma-S, binding against human Opioid receptor like 1 | J Med Chem 45: 5353-7 (2002) BindingDB Entry DOI: 10.7270/Q2VT1ST9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cells | J Med Chem 43: 4667-77 (2001) BindingDB Entry DOI: 10.7270/Q2NK3D9H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 102 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Stimulation of [35S]GTP-gamma-S, binding using COS-human Opioid receptor mu 1 membranes | J Med Chem 45: 5353-7 (2002) BindingDB Entry DOI: 10.7270/Q2VT1ST9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 103 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]diprenorphine (0.33 nM) binding from human Opioid receptor mu 1 expressed in CHO-K1 cells. | J Med Chem 43: 4667-77 (2001) BindingDB Entry DOI: 10.7270/Q2NK3D9H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 1.06E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Stimulation of [35S]GTP-gamma-S, binding using COS-human Opioid receptor kappa 1 membranes | J Med Chem 45: 5353-7 (2002) BindingDB Entry DOI: 10.7270/Q2VT1ST9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 1.06E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]naltrindole (0.55 nM) binding from human Opioid receptor kappa 1 | J Med Chem 43: 4667-77 (2001) BindingDB Entry DOI: 10.7270/Q2NK3D9H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 8.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Binding affinity for opioid receptor type, human Opioid receptor delta 1 expressed in membrane homogenates of COS-1 or CHO cells | J Med Chem 45: 5353-7 (2002) BindingDB Entry DOI: 10.7270/Q2VT1ST9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 8.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]naltrindole (0.55 nM) binding from human Opioid receptor delta 1 expressed in CHO-K1 cells. | J Med Chem 43: 4667-77 (2001) BindingDB Entry DOI: 10.7270/Q2NK3D9H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Superiore di Sanit£ Curated by ChEMBL | Assay Description Displacement of [3H]-naltrindole from human mu opioid receptor expressed in CHO cells after 2 hrs by scintillation counting | Eur J Med Chem 46: 1207-21 (2011) Article DOI: 10.1016/j.ejmech.2011.01.040 BindingDB Entry DOI: 10.7270/Q2NK3FBC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Superiore di Sanit£ Curated by ChEMBL | Assay Description Displacement of [3H]-naltrindole from human kappa opioid receptor expressed in human HEK293 cells after 2 hrs by scintillation counting | Eur J Med Chem 46: 1207-21 (2011) Article DOI: 10.1016/j.ejmech.2011.01.040 BindingDB Entry DOI: 10.7270/Q2NK3FBC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nociceptin receptor (Homo sapiens (Human)) | BDBM50094634 (CHEMBL140979 | N-(4-Amino-2-methyl-quinolin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Superiore di Sanit£ Curated by ChEMBL | Assay Description Displacement of [125I]Tyr14-nociceptin from human NOP receptor expressed in human HEK293 cells after 2 hrs by scintillation counting | Eur J Med Chem 46: 1207-21 (2011) Article DOI: 10.1016/j.ejmech.2011.01.040 BindingDB Entry DOI: 10.7270/Q2NK3FBC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||