

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50098634 1-Cyclooctylmethyl-1-methyl-4-[(9H-xanthene-9-carbonyl)-amino]-piperidinium; iodide::CHEMBL34165
SMILES: C[N+]1(CC2CCCCCCC2)CCC(CC1)NC(=O)C1c2ccccc2Oc2ccccc12
InChI Key: InChIKey=XRGRFIJPNWJGGI-UHFFFAOYSA-O
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C-C chemokine receptor type 1 (Homo sapiens (Human)) | BDBM50098634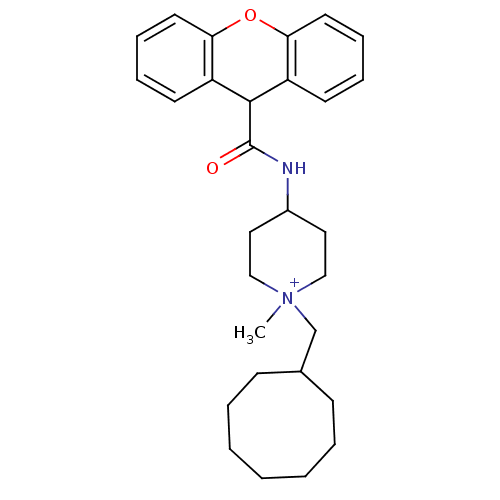 (1-Cyclooctylmethyl-1-methyl-4-[(9H-xanthene-9-carb...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against [125I]-MIP-1 alpha binding to human CCR1 receptors. | J Med Chem 44: 1429-35 (2001) BindingDB Entry DOI: 10.7270/Q2G73D0G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| C-C chemokine receptor type 1 (Mus musculus) | BDBM50098634 (1-Cyclooctylmethyl-1-methyl-4-[(9H-xanthene-9-carb...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Banyu Tsukuba Research Institute in collaboration with Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against [125I]-MIP-1 alpha binding to mouse CCR1 receptors. | J Med Chem 44: 1429-35 (2001) BindingDB Entry DOI: 10.7270/Q2G73D0G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||