 Found 14 hits for monomerid = 50101828
Found 14 hits for monomerid = 50101828 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostanoid EP3 Receptor
(Mus musculus (Mouse)) | BDBM50101828
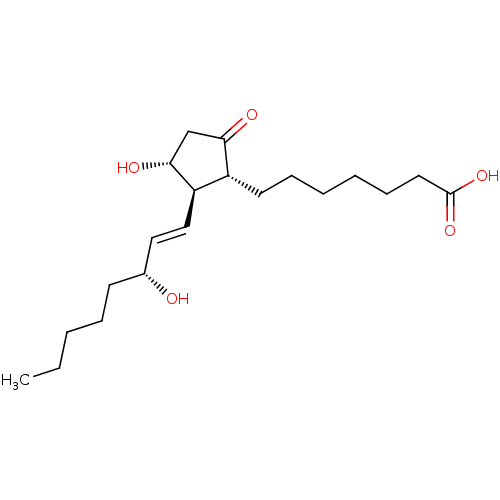
(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
Br J Pharmacol 122: 217-24 (1997)
Article DOI: 10.1038/sj.bjp.0701367
BindingDB Entry DOI: 10.7270/Q26M35CT |
More data for this
Ligand-Target Pair | |
PTGER3
(BOVINE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
PTGER4
(Mus musculus (Mouse)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
Br J Pharmacol 122: 217-24 (1997)
Article DOI: 10.1038/sj.bjp.0701367
BindingDB Entry DOI: 10.7270/Q26M35CT |
More data for this
Ligand-Target Pair | |
PTGER3
(BOVINE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Prostanoid EP2 Receptor
(Mus musculus (Mouse)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
Br J Pharmacol 122: 217-24 (1997)
Article DOI: 10.1038/sj.bjp.0701367
BindingDB Entry DOI: 10.7270/Q26M35CT |
More data for this
Ligand-Target Pair | |
PTGIR
(MOUSE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
Br J Pharmacol 122: 217-24 (1997)
Article DOI: 10.1038/sj.bjp.0701367
BindingDB Entry DOI: 10.7270/Q26M35CT |
More data for this
Ligand-Target Pair | |
Prostanoid EP1 Receptor
(Mus musculus (Mouse)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
Br J Pharmacol 122: 217-24 (1997)
Article DOI: 10.1038/sj.bjp.0701367
BindingDB Entry DOI: 10.7270/Q26M35CT |
More data for this
Ligand-Target Pair | |
Prostanoid IP receptor
(Homo sapiens (Human)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 137 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Royal Postgraduate Medical School
Curated by PDSP Ki Database
| |
Br J Pharmacol 72: 435-41 (1981)
Article DOI: 10.1111/j.1476-5381.1981.tb10994.x
BindingDB Entry DOI: 10.7270/Q2HM56XZ |
More data for this
Ligand-Target Pair | |
PTGIR
(MOUSE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 137 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Royal Postgraduate Medical School
Curated by PDSP Ki Database
| |
Biochem Pharmacol 30: 2041-4 (1981)
Article DOI: 10.1016/0006-2952(81)90220-3
BindingDB Entry DOI: 10.7270/Q2668BPP |
More data for this
Ligand-Target Pair | |
PTGDR
(RAT) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.76E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by PDSP Ki Database
| |
J Biol Chem 257: 13570-5 (1982)
BindingDB Entry DOI: 10.7270/Q22F7KXH |
More data for this
Ligand-Target Pair | |
TBXA2R
(MOUSE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Edinburgh
Curated by PDSP Ki Database
| |
Br J Pharmacol 79: 953-64 (1983)
Article DOI: 10.1111/j.1476-5381.1983.tb10541.x
BindingDB Entry DOI: 10.7270/Q2SX6BPF |
More data for this
Ligand-Target Pair | |
PTGFR
(BOVINE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | n/a | n/a | 0.220 | n/a | n/a | n/a | 37 |
Cayman Chemical Company, Inc.
US Patent
| Assay Description
1. Seed cells on an EP2 or EP4 STEP plate at a density of 40,000-80,000 cells/well in 200 uL of reduced serum medium containing 0.5% FBS. Place the p... |
US Patent US9180116 (2015)
BindingDB Entry DOI: 10.7270/Q2QC029S |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | n/a | n/a | 16.5 | n/a | n/a | n/a | 37 |
Cayman Chemical Company, Inc.
US Patent
| Assay Description
1. Seed cells on an EP2 or EP4 STEP plate at a density of 40,000-80,000 cells/well in 200 ul of reduced serum medium containing 0.5% FBS. Place the p... |
US Patent US9180116 (2015)
BindingDB Entry DOI: 10.7270/Q2QC029S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data