

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50102271 1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-triaza-cyclopenta[a]naphthalen-5-yl)-ethyl]-thiourea::CHEMBL107535
SMILES: Brc1ccc(NC(=S)NCCn2c3cccnc3n3cccc3c2=O)nc1
InChI Key: InChIKey=XRESPDQVPMSLGC-UHFFFAOYSA-N
Data: 6 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271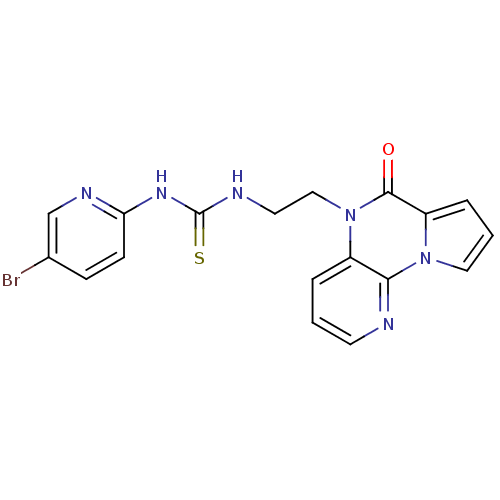 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 wild-type RT | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 Mutant HIV-1 RT enzymes containing the single amino acid substitution V106A | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 95 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 Mutant HIV-1 RT enzymes containing the single amino acid substitution L1001 | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 Mutant HIV-1 RT enzymes containing the single amino acid substitution Y188L | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 Mutant HIV-1 RT enzymes containing the single amino acid substitution K103N | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50102271 (1-(5-Bromo-pyridin-2-yl)-3-[2-(4-oxo-4H-5,9,9b-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of HIV-1 Mutant HIV-1 RT enzymes containing the single amino acid substitution Y181I | J Med Chem 44: 305-15 (2001) BindingDB Entry DOI: 10.7270/Q2HX1DD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||