

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50102295 4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethyl-pyridin-2-ylazo)-benzoic acid::CHEMBL119235
SMILES: Cc1nc(N=Nc2ccc(cc2)C(O)=O)c(COP(O)(O)=O)c(C=O)c1O
InChI Key: InChIKey=NPBWMMRUXMTIRC-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P2Y purinoceptor 1 (Meleagris gallopavo) | BDBM50102295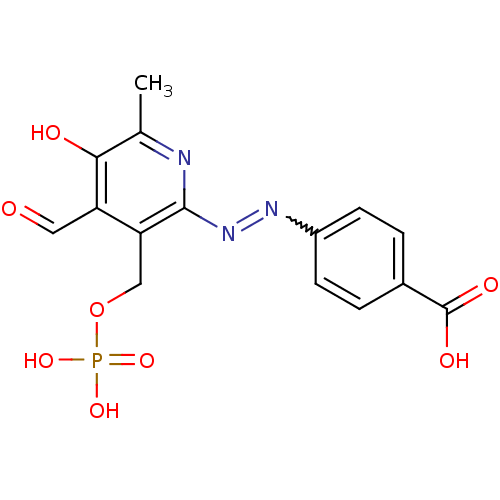 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of 10 nM 2-MeSADP-stimulated phospholipase C in turkey erythrocyte membranes using [3H]-inositol | J Med Chem 44: 340-9 (2001) BindingDB Entry DOI: 10.7270/Q2FN15HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 1 (RAT) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 9.40 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of inward ion current elicited by ATP at P2X1 receptor expressed in Xenopus oocytes | J Med Chem 44: 340-9 (2001) BindingDB Entry DOI: 10.7270/Q2FN15HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic, P2X2 (RAT) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 1.19E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of inward ion current elicited by ATP at P2X2 receptor expressed in Xenopus oocytes | J Med Chem 44: 340-9 (2001) BindingDB Entry DOI: 10.7270/Q2FN15HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 3 (RAT) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of inward ion current elicited by ATP was determined at recombinant P2X3 receptor expressed in Xenopus oocytes | J Med Chem 44: 340-9 (2001) BindingDB Entry DOI: 10.7270/Q2FN15HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 14-3-3 protein zeta/delta (Homo sapiens) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eindhoven University of Technology Curated by ChEMBL | Assay Description Binding affinity to GST-tagged 14-3-3-zeta (unknown origin) expressed in Escherichia coli BL21(DE3) assessed as inhibition of interaction with PRAS40... | J Med Chem 61: 3755-3778 (2018) Article DOI: 10.1021/acs.jmedchem.7b00574 BindingDB Entry DOI: 10.7270/Q2RB7781 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| P2X purinoceptor 1 (Mus musculus) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology (GIST) Curated by ChEMBL | Assay Description Antagonist activity at recombinant mouse P2X1 receptor expressed in Xenopus oocytes assessed as inhibition of ATP-induced ion current preincubated fo... | Eur J Med Chem 70: 811-30 (2013) Article DOI: 10.1016/j.ejmech.2013.10.026 BindingDB Entry DOI: 10.7270/Q2M61MQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 7 (Homo sapiens (Human)) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology Curated by ChEMBL | Assay Description Antagonist activity at P2X7 receptor (unknown origin) | Bioorg Med Chem 21: 2643-50 (2013) Article DOI: 10.1016/j.bmc.2013.01.073 BindingDB Entry DOI: 10.7270/Q2DF6SK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 14-3-3 protein zeta/delta (Homo sapiens) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB PubMed | n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rheinische Friedrich-Wilhelms-Universit£t Curated by ChEMBL | Assay Description Inhibition of PRAS40 binding to human GST-tagged 14-3-3 protein zeta/delta expressed in Escherichia coli BL21(DE3) by ELISA | J Med Chem 61: 1276-1284 (2018) BindingDB Entry DOI: 10.7270/Q2B27XQN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein, gamma polypeptide (Homo sapiens (Human)) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eindhoven University of Technology Curated by ChEMBL | Assay Description Binding affinity to GST-tagged 14-3-3-gamma (unknown origin) expressed in Escherichia coli BL21(DE3) assessed as inhibition of interaction with PRAS4... | J Med Chem 61: 3755-3778 (2018) Article DOI: 10.1021/acs.jmedchem.7b00574 BindingDB Entry DOI: 10.7270/Q2RB7781 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 1 (RAT) | BDBM50102295 (4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology (GIST) Curated by ChEMBL | Assay Description Antagonist activity at recombinant rat P2X1 receptor expressed in Xenopus oocytes assessed as ion flux stimulation | Eur J Med Chem 70: 811-30 (2013) Article DOI: 10.1016/j.ejmech.2013.10.026 BindingDB Entry DOI: 10.7270/Q2M61MQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||