 Found 13 hits for monomerid = 50104501
Found 13 hits for monomerid = 50104501 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Peptide deformylase
(Escherichia coli) | BDBM50104501
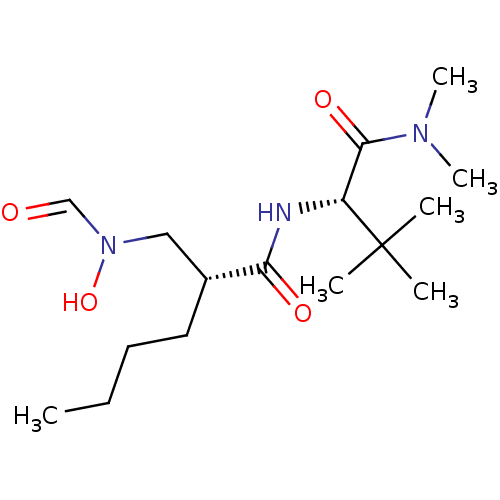
((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University
Curated by ChEMBL
| Assay Description
Inhibitory effect against E. coli peptide deformylase (PDF) by DPPI assay |
J Med Chem 47: 4941-9 (2004)
Article DOI: 10.1021/jm049592c
BindingDB Entry DOI: 10.7270/Q2WD401S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptide deformylase
(Escherichia coli) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against Escherichia coli peptide deformylase (PDF) Nickel containing enzyme |
Bioorg Med Chem Lett 14: 59-62 (2003)
BindingDB Entry DOI: 10.7270/Q2T43SHT |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Compound was tested for its inhibitory activity against Angiotensin I converting enzyme |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
The compound was evaluated in vitro for the inhibition of Neutral endopeptidase |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloproteinase-3 |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Peptide deformylase mitochondrial
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Antibacterial activity of the compound against E. coli Peptide deformylase. Ni |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Peptide deformylase
(Escherichia coli) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition concentration against Escherichia coli peptide deformylase. |
Bioorg Med Chem Lett 13: 2715-8 (2003)
BindingDB Entry DOI: 10.7270/Q2VQ336P |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Matrix metalloproteinase-7 (MMP7)
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloproteinase-7 |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition of human Matrix metalloproteinase-1 |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |
Peptide deformylase mitochondrial
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Compound was evaluated for inhibition of peptide deformylase, PDF.Ni of Escherichia coli |
Bioorg Med Chem Lett 12: 3595-9 (2002)
BindingDB Entry DOI: 10.7270/Q2FJ2G4H |
More data for this
Ligand-Target Pair | |
Peptide deformylase mitochondrial
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition against Escherichia coli peptide deformylase |
Bioorg Med Chem Lett 13: 2709-13 (2003)
BindingDB Entry DOI: 10.7270/Q2XW4J6S |
More data for this
Ligand-Target Pair | |
Peptide deformylase
(Escherichia coli) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition concentration against Escherichia coli peptide deformylase. |
Bioorg Med Chem Lett 13: 2715-8 (2003)
BindingDB Entry DOI: 10.7270/Q2VQ336P |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50104501

((R)-2-[(Formyl-hydroxy-amino)-methyl]-hexanoic aci...)Show SMILES CCCC[C@H](CN(O)C=O)C(=O)N[C@H](C(=O)N(C)C)C(C)(C)C Show InChI InChI=1S/C16H31N3O4/c1-7-8-9-12(10-19(23)11-20)14(21)17-13(16(2,3)4)15(22)18(5)6/h11-13,23H,7-10H2,1-6H3,(H,17,21)/t12-,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
British Biotech Pharmaceuticals Limited
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloproteinase-2 |
Bioorg Med Chem Lett 11: 2585-8 (2001)
BindingDB Entry DOI: 10.7270/Q2SN0886 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data