

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50110768 5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-ethyl-pyrimidine-2,4-diamine::CHEMBL22129
SMILES: CCc1nc(N)nc(N)c1-c1ccc(OCc2ccc(Cl)cc2Cl)c(OC)c1
InChI Key: InChIKey=COLNPIYVAKVLJG-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 (Plasmodium falciparum (isolate K1 / Thailand)) | BDBM50110768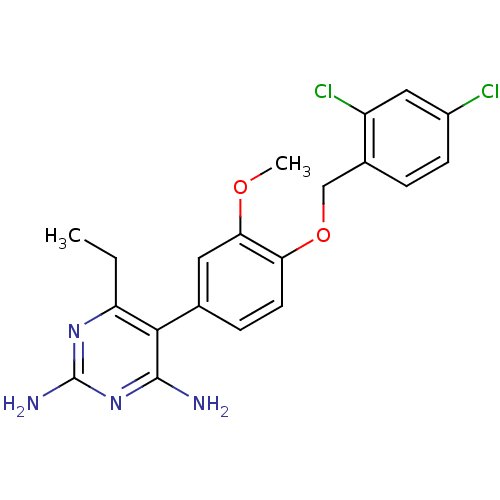 (5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Genetic Engineering and Biotechnology Curated by ChEMBL | Assay Description Inhibition of the wild-type dihydrofolate reductase (DHFR) | J Med Chem 45: 1244-52 (2002) BindingDB Entry DOI: 10.7270/Q2Z89BQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 (Plasmodium falciparum (isolate K1 / Thailand)) | BDBM50110768 (5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Modena e Reggio Emilia Curated by ChEMBL | Assay Description Inhibition constant against Plasmodium falciparum dihydrofolate reductase | J Med Chem 47: 4258-67 (2004) Checked by Author Article DOI: 10.1021/jm040769c BindingDB Entry DOI: 10.7270/Q2HH6JKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 (Plasmodium falciparum (isolate K1 / Thailand)) | BDBM50110768 (5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 20.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Genetic Engineering and Biotechnology Curated by ChEMBL | Assay Description Inhibition of the S108N mutant of dihydrofolate reductase (DHFR) | J Med Chem 45: 1244-52 (2002) BindingDB Entry DOI: 10.7270/Q2Z89BQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 (Plasmodium falciparum (isolate K1 / Thailand)) | BDBM50110768 (5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 42.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Genetic Engineering and Biotechnology Curated by ChEMBL | Assay Description Inhibition of the C59R+S108N mutant of dihydrofolate reductase (DHFR) | J Med Chem 45: 1244-52 (2002) BindingDB Entry DOI: 10.7270/Q2Z89BQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 (Plasmodium falciparum (isolate K1 / Thailand)) | BDBM50110768 (5-[4-(2,4-Dichloro-benzyloxy)-3-methoxy-phenyl]-6-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Genetic Engineering and Biotechnology Curated by ChEMBL | Assay Description Antiplasmodial activity (IC50) against Plasmodium falciparum Clone with mutant enzyme C59R+S108N- pfDihydrofolate reductase (K1CB1) | J Med Chem 45: 1244-52 (2002) BindingDB Entry DOI: 10.7270/Q2Z89BQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||