

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50115150 2-Amino-6-methylaminomethyl-5,6,7,8-tetrahydro-3H-pteridin-4-one::CHEMBL420453
SMILES: CNCC1CNc2nc(N)[nH]c(=O)c2N1
InChI Key: InChIKey=LHIQNSIRCHEIBK-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, brain (Homo sapiens (Human)) | BDBM50115150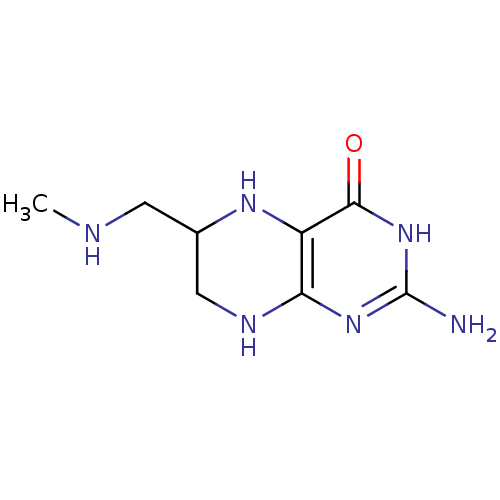 (2-Amino-6-methylaminomethyl-5,6,7,8-tetrahydro-3H-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharma Curated by ChEMBL | Assay Description Inhibitory activity against human neuronal nitric oxide synthase (NOS-I) | J Med Chem 45: 2923-41 (2002) BindingDB Entry DOI: 10.7270/Q2571BBK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50115150 (2-Amino-6-methylaminomethyl-5,6,7,8-tetrahydro-3H-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Curated by ChEMBL | Assay Description Inhibitory activity against human Nitric oxide synthase-III with 2 uM H4Bip for 30 min | J Med Chem 48: 4783-92 (2005) Article DOI: 10.1021/jm050007x BindingDB Entry DOI: 10.7270/Q28W3CTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50115150 (2-Amino-6-methylaminomethyl-5,6,7,8-tetrahydro-3H-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.06E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Curated by ChEMBL | Assay Description Inhibitory activity against human Nitric oxide synthase-II with 2 uM H4Bip for 30 min | J Med Chem 48: 4783-92 (2005) Article DOI: 10.1021/jm050007x BindingDB Entry DOI: 10.7270/Q28W3CTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, brain (Homo sapiens (Human)) | BDBM50115150 (2-Amino-6-methylaminomethyl-5,6,7,8-tetrahydro-3H-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Curated by ChEMBL | Assay Description Inhibitory activity against human Nitric oxide synthase-I with 2 uM H4Bip for 30 min | J Med Chem 48: 4783-92 (2005) Article DOI: 10.1021/jm050007x BindingDB Entry DOI: 10.7270/Q28W3CTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||