

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50117778 2-[2-(4-Fluoro-benzylsulfanyl)-5-(1-methyl-1H-pyrazol-4-ylmethyl)-4-oxo-4H-pyrimidin-1-yl]-N-(2-piperidin-1-yl-ethyl)-N-(4'-trifluoromethyl-biphenyl-4-ylmethyl)-acetamide::CHEMBL314768
SMILES: Cn1cc(Cc2cn(CC(=O)N(CCN3CCCCC3)Cc3ccc(cc3)-c3ccc(cc3)C(F)(F)F)c(SCc3ccc(F)cc3)nc2=O)cn1
InChI Key: InChIKey=RIDDCLHNDOXTES-UHFFFAOYSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50117778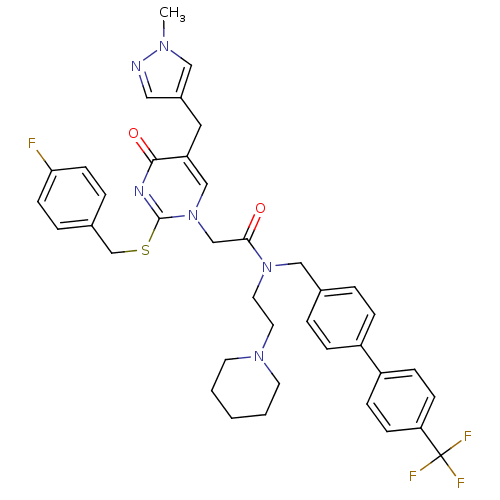 (2-[2-(4-Fluoro-benzylsulfanyl)-5-(1-methyl-1H-pyra...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of cytochrome P450 3A4 | Bioorg Med Chem Lett 12: 2603-6 (2002) BindingDB Entry DOI: 10.7270/Q2G44PNN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| LDL-associated phospholipase A2 (Homo sapiens (Human)) | BDBM50117778 (2-[2-(4-Fluoro-benzylsulfanyl)-5-(1-methyl-1H-pyra...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibitory concentration against Lipoprotein-Associated Phospholipase A2 (Lp-PLA2) was estimated | Bioorg Med Chem Lett 12: 2603-6 (2002) BindingDB Entry DOI: 10.7270/Q2G44PNN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||