

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50127480 2-(1,1-Dioxo-7-phenyl-1,3,4,5-tetrahydro-1lambda*6*-benzo[f][1,2,5]thiadiazepin-2-yl)-N-hydroxy-propionamide::CHEMBL298635
SMILES: C[C@@H](N1CCNc2cc(ccc2S1(=O)=O)-c1ccccc1)C(=O)NO
InChI Key: InChIKey=BNTJDVFEUJBXMT-GFCCVEGCSA-N
Data: 4 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50127480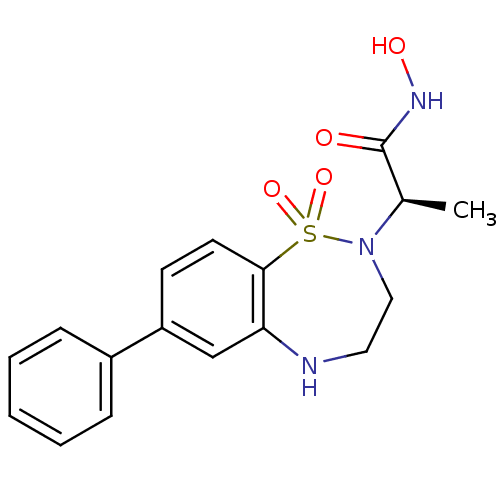 (2-(1,1-Dioxo-7-phenyl-1,3,4,5-tetrahydro-1lambda*6...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description In vitro inhibitory activity against broad spectrum matrix metalloprotease-2 (MMP-2) | J Med Chem 46: 1811-23 (2003) Article DOI: 10.1021/jm020475w BindingDB Entry DOI: 10.7270/Q2TQ60W7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50127480 (2-(1,1-Dioxo-7-phenyl-1,3,4,5-tetrahydro-1lambda*6...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description In vitro inhibitory activity against broad spectrum matrix metalloprotease-9 (MMP-9) | J Med Chem 46: 1811-23 (2003) Article DOI: 10.1021/jm020475w BindingDB Entry DOI: 10.7270/Q2TQ60W7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50127480 (2-(1,1-Dioxo-7-phenyl-1,3,4,5-tetrahydro-1lambda*6...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 272 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description In vitro inhibitory activity against broad spectrum matrix metalloprotease-1 (MMP-1) | J Med Chem 46: 1811-23 (2003) Article DOI: 10.1021/jm020475w BindingDB Entry DOI: 10.7270/Q2TQ60W7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Disintegrin and metalloproteinase domain-containing protein 17 (Homo sapiens (Human)) | BDBM50127480 (2-(1,1-Dioxo-7-phenyl-1,3,4,5-tetrahydro-1lambda*6...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description In vitro inhibitory activity against porcine tumor necrosis factor alpha converting enzyme (pTACE) | J Med Chem 46: 1811-23 (2003) Article DOI: 10.1021/jm020475w BindingDB Entry DOI: 10.7270/Q2TQ60W7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||