

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CCCC[C@]1(CC)CS(=O)(=O)c2cc(OC)c(OC)cc2[C@H](N1)c1ccccc1
InChI Key: InChIKey=CKFWDLFFXXVSBJ-DHIUTWEWSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ileal sodium/bile acid cotransporter (Homo sapiens (Human)) | BDBM50134411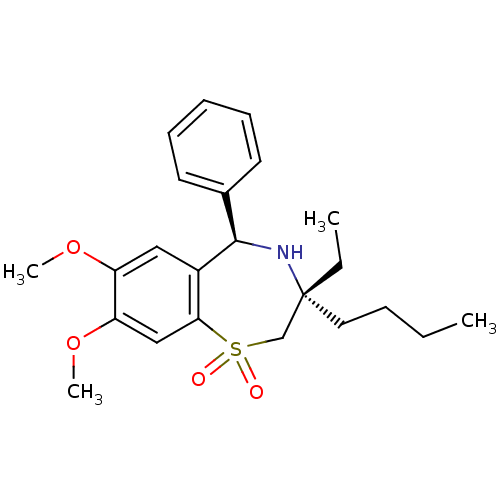 ((7R,9R)-7-Butyl-7-ethyl-2,3-dimethoxy-9-phenyl-6,7...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description In vitro inhibitory activity against uptake of [14C]-taurocholate in baby hamster kidney cells transfected with cDNA from human Apical Sodium-Codepen... | Bioorg Med Chem Lett 13: 3727-30 (2003) BindingDB Entry DOI: 10.7270/Q2M044T4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ileal sodium/bile acid cotransporter (Rattus norvegicus) | BDBM50134411 ((7R,9R)-7-Butyl-7-ethyl-2,3-dimethoxy-9-phenyl-6,7...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research& Development Curated by ChEMBL | Assay Description Inhibition of rat ASBT expressed in HEK293 cells assessed as inhibition of [3H]-taurocholate uptake after 90 mins by scintillation counting analysis | J Med Chem 56: 5094-114 (2013) Article DOI: 10.1021/jm400459m BindingDB Entry DOI: 10.7270/Q2MC91D6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ileal sodium/bile acid cotransporter (Mus musculus) | BDBM50134411 ((7R,9R)-7-Butyl-7-ethyl-2,3-dimethoxy-9-phenyl-6,7...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research& Development Curated by ChEMBL | Assay Description Inhibition of mouse ASBT expressed in HEK293 cells assessed as inhibition of [3H]-taurocholate uptake after 90 mins by scintillation counting analysi... | J Med Chem 56: 5094-114 (2013) Article DOI: 10.1021/jm400459m BindingDB Entry DOI: 10.7270/Q2MC91D6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ileal sodium/bile acid cotransporter (Homo sapiens (Human)) | BDBM50134411 ((7R,9R)-7-Butyl-7-ethyl-2,3-dimethoxy-9-phenyl-6,7...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research& Development Curated by ChEMBL | Assay Description Inhibition of human ASBT expressed in HEK293 cells assessed as inhibition of [3H]-taurocholate uptake after 90 mins by scintillation counting analysi... | J Med Chem 56: 5094-114 (2013) Article DOI: 10.1021/jm400459m BindingDB Entry DOI: 10.7270/Q2MC91D6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||