

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50138474 (1R,3R,5S)-3-Benzo[b]thiophen-3-yl-8-((S)-8-methoxy-2,3-dihydro-benzo[1,4]dioxin-2-ylmethyl)-8-aza-bicyclo[3.2.1]octan-3-ol::CHEMBL148860
SMILES: COc1cccc2OC[C@H](CN3[C@H]4CC[C@@H]3C[C@](O)(C4)c3csc4ccccc34)Oc12
InChI Key: InChIKey=WHNFSMFOYMLNKR-BGWODGLRSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 1A (Homo sapiens (Human)) | BDBM50138474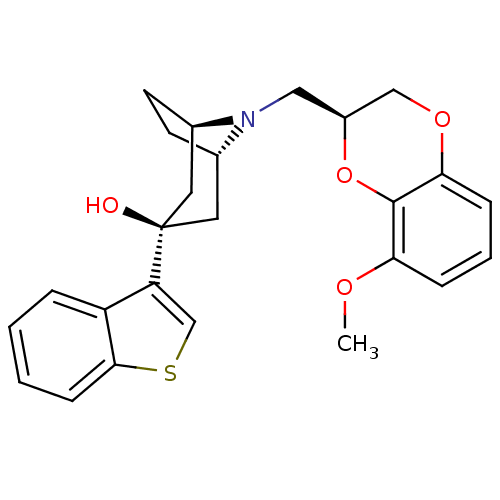 ((1R,3R,5S)-3-Benzo[b]thiophen-3-yl-8-((S)-8-methox...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Displacement of [3H]8-OH-DPAT from human 5-hydroxytryptamine 1A receptor expressed in CHO cells | Bioorg Med Chem Lett 14: 515-8 (2003) BindingDB Entry DOI: 10.7270/Q247499J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor (Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50138474 ((1R,3R,5S)-3-Benzo[b]thiophen-3-yl-8-((S)-8-methox...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 106 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Agonsitic activity of compound towards 5-hydroxytryptamine 1A receptor was evaluated by [35S]-GTP-gammaS, stimulated cAMP assay | Bioorg Med Chem Lett 14: 515-8 (2003) BindingDB Entry DOI: 10.7270/Q247499J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Homo sapiens (Human)) | BDBM50138474 ((1R,3R,5S)-3-Benzo[b]thiophen-3-yl-8-((S)-8-methox...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Agonistic activity of compound towards 5-hydroxytryptamine 1A receptor was evaluated by forskolin stimulated cAMP assay | Bioorg Med Chem Lett 14: 515-8 (2003) BindingDB Entry DOI: 10.7270/Q247499J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Homo sapiens (Human)) | BDBM50138474 ((1R,3R,5S)-3-Benzo[b]thiophen-3-yl-8-((S)-8-methox...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Agonsitic activity of compound towards 5-hydroxytryptamine 1A receptor was evaluated by [35S]-GTP-gammaS, stimulated cAMP assay | Bioorg Med Chem Lett 14: 515-8 (2003) BindingDB Entry DOI: 10.7270/Q247499J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||