 Found 5 hits for monomerid = 50144936
Found 5 hits for monomerid = 50144936 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosylhomocysteinase
(Mus musculus) | BDBM50144936
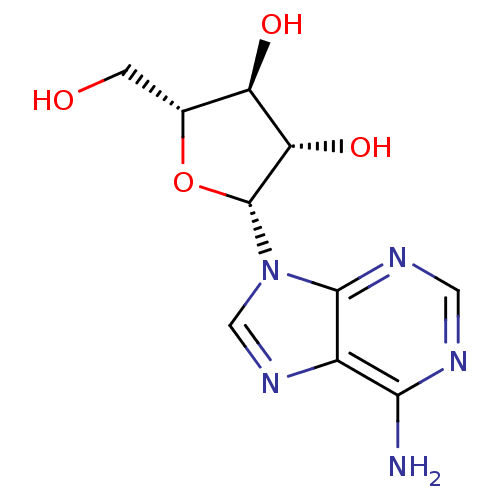
(CHEMBL1090 | VIDARABINE | adenine arabinoside)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7+,10-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute
Curated by ChEMBL
| Assay Description
Activity determined in mouse liver S-adenosyl-L-homocysteine hydrolase and expressed as KI values. |
J Med Chem 34: 647-56 (1991)
BindingDB Entry DOI: 10.7270/Q25M6695 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50144936

(CHEMBL1090 | VIDARABINE | adenine arabinoside)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7+,10-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 62 | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Qu£mica M£dica (C.S.I.C.)
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 from human Caco2 cells |
J Med Chem 53: 559-72 (2010)
Article DOI: 10.1021/jm901590f
BindingDB Entry DOI: 10.7270/Q2P55NMC |
More data for this
Ligand-Target Pair | |
Hepatitis C virus NS5B RNA-dependent RNA polymerase
(Hepatitis C virus) | BDBM50144936

(CHEMBL1090 | VIDARABINE | adenine arabinoside)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7+,10-/m1/s1 | UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Isis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition HCV NS5B-mediated RNA synthesis |
J Med Chem 47: 2283-95 (2004)
Article DOI: 10.1021/jm030424e
BindingDB Entry DOI: 10.7270/Q2QZ2BQ3 |
More data for this
Ligand-Target Pair | |
Hepatitis C virus NS5B RNA-dependent RNA polymerase
(Hepatitis C virus) | BDBM50144936

(CHEMBL1090 | VIDARABINE | adenine arabinoside)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7+,10-/m1/s1 | UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a |
Isis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition HCV RNA replication |
J Med Chem 47: 2283-95 (2004)
Article DOI: 10.1021/jm030424e
BindingDB Entry DOI: 10.7270/Q2QZ2BQ3 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM50144936

(CHEMBL1090 | VIDARABINE | adenine arabinoside)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7+,10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a |
Broad Institute
Curated by PubChem BioAssay
| Assay Description
Keywords: GSK3beta, dose response, kinase, inhibition, HTS Assay Overview: The glycogen synthase kinase-3 beta (GSK-3b) is a known master regulator f... |
PubChem Bioassay (2010)
BindingDB Entry DOI: 10.7270/Q2TX3CTT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data