

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50145571 1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1-[4-(4-methyl-piperazin-1-yl)-cyclohexyl]-1H-pyrazolo[3,4-d]pyrimidin-3-yl}-2-methoxy-phenyl)-amide::A-420983::CHEMBL314627::N-(4-{4-AMINO-1-[4-(4-METHYLPIPERAZIN-1-YL)-TRANS-CYCLOHEXYL]-1H-PYRAZOLO[3,4-D]PYRIMIDIN-3-YL}-2-METHOXYPHENYL)-1-METHYL-1H-INDOLE-2-CARBOXAMIDE::US10688093, Compound A-420983 (Abbott)
SMILES: COc1cc(ccc1NC(=O)c1cc2ccccc2n1C)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12
InChI Key: InChIKey=STVKLDUINKMZFE-RQNOJGIXSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tyrosine-protein kinase Lck (Homo sapiens (Human)) | BDBM50145571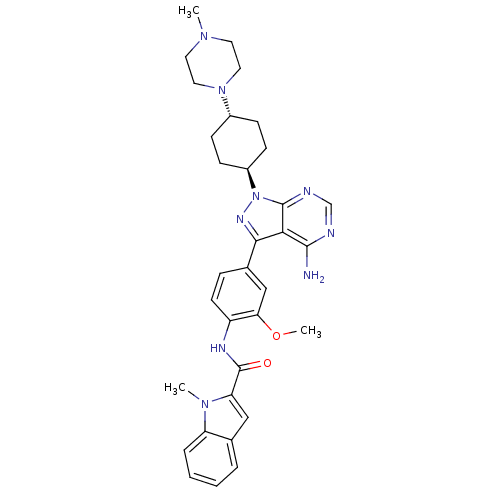 (1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center Curated by ChEMBL | Assay Description Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide | Bioorg Med Chem Lett 14: 2613-6 (2004) Article DOI: 10.1016/j.bmcl.2004.02.101 BindingDB Entry DOI: 10.7270/Q2M61JQP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM50145571 (1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center Curated by ChEMBL | Assay Description Inhibition of vascular endothelial growth factor receptor 2 activity with 1mM ATP and biotinylated lck peptide | Bioorg Med Chem Lett 14: 2613-6 (2004) Article DOI: 10.1016/j.bmcl.2004.02.101 BindingDB Entry DOI: 10.7270/Q2M61JQP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase Fyn (Homo sapiens (Human)) | BDBM50145571 (1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA US Patent | Assay Description The assay procedure determines the IC50 of each potential FYN kinase inhibitor by measuring the enzyme catalyzed ATP-dependent phosphorylation of the... | US Patent US10688093 (2020) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiopoietin-1 receptor (Homo sapiens (Human)) | BDBM50145571 (1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center Curated by ChEMBL | Assay Description Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide | Bioorg Med Chem Lett 14: 2613-6 (2004) Article DOI: 10.1016/j.bmcl.2004.02.101 BindingDB Entry DOI: 10.7270/Q2M61JQP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM50145571 (1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center Curated by ChEMBL | Assay Description Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide | Bioorg Med Chem Lett 14: 2613-6 (2004) Article DOI: 10.1016/j.bmcl.2004.02.101 BindingDB Entry DOI: 10.7270/Q2M61JQP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||