

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50145862 CHEMBL3765171
SMILES: Cc1c(N[C@@H]2CCC[C@]2(C)O)ccc(C#N)c1Cl
InChI Key: InChIKey=CXFSVKGROITHRY-OCCSQVGLSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Androgen Receptor (Homo sapiens (Human)) | BDBM50145862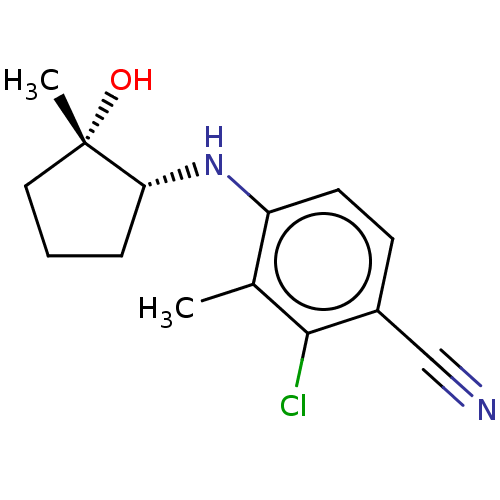 (CHEMBL3765171) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-methyltrienolone from androgen receptor (unknown origin) expressed in HEK293 cell lysate incubated overnight by microbeta scinti... | J Med Chem 59: 750-5 (2016) BindingDB Entry DOI: 10.7270/Q2H70HQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50145862 (CHEMBL3765171) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 448 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-methyltrienolone from progesterone receptor (unknown origin) expressed in HEK293 cell lysate incubated overnight by microbeta sc... | J Med Chem 59: 750-5 (2016) BindingDB Entry DOI: 10.7270/Q2H70HQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glucocorticoid receptor (Homo sapiens (Human)) | BDBM50145862 (CHEMBL3765171) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 462 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-dexamethasone from glucocorticoid receptor (unknown origin) expressed in HEK293 cell lysate incubated overnight by microbeta sci... | J Med Chem 59: 750-5 (2016) BindingDB Entry DOI: 10.7270/Q2H70HQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mineralocorticoid receptor (Homo sapiens (Human)) | BDBM50145862 (CHEMBL3765171) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-aldosterone from mineralocorticoid receptor (unknown origin) expressed in HEK293 cell lysate incubated overnight by microbeta sc... | J Med Chem 59: 750-5 (2016) BindingDB Entry DOI: 10.7270/Q2H70HQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen Receptor (Homo sapiens (Human)) | BDBM50145862 (CHEMBL3765171) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 0.0398 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at human androgen receptor expressed in mouse C2C12 cells cotransfected with GRE/ARE reporter plasmid assessed as receptor transacti... | J Med Chem 59: 750-5 (2016) BindingDB Entry DOI: 10.7270/Q2H70HQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||