

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50153093 CHEMBL3775668
SMILES: O=c1[nH]cnc2c(nccc12)-n1cc(CCN2CCC(CC2)c2cccs2)cn1
InChI Key: InChIKey=XIVQRYFYMNIDET-UHFFFAOYSA-N
Data: 6 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lysine-specific demethylase 3A (Homo sapiens (Human)) | BDBM50153093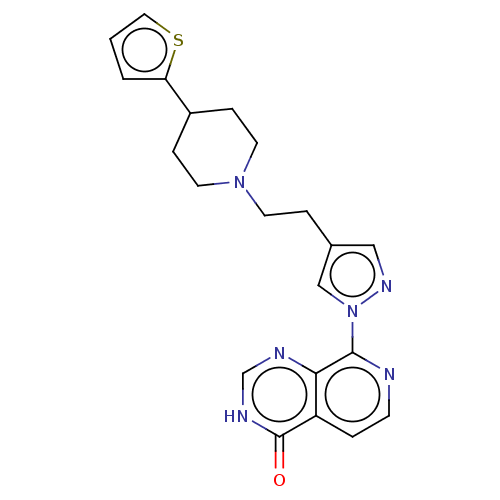 (CHEMBL3775668) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of KDM3A (unknown origin) using biotin-H3K9me2 (1 to 21 residues) as substrate preincubated for 15 mins followed by substrate addition mea... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific demethylase 5C (Homo sapiens (Human)) | BDBM50153093 (CHEMBL3775668) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of KDM5C (unknown origin) using biotin-H3K4me3 as substrate preincubated for 15 mins followed by substrate addition measured after 20 mins... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific demethylase 5B (Homo sapiens (Human)) | BDBM50153093 (CHEMBL3775668) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of KDM5B (unknown origin) using biotin-H3K4me3 as substrate preincubated for 15 mins followed by substrate addition measured after 20 mins... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific demethylase 4B (Homo sapiens (Human)) | BDBM50153093 (CHEMBL3775668) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal GST-tagged KDM4B (1 to 500 residues) expressed in baculovirus infected sf9 cells using biotin-H3K9me3 as s... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Homo sapiens lysine demethylase 2A (KDM2A) (Homo sapiens (Human)) | BDBM50153093 (CHEMBL3775668) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of KDM2A (unknown origin) using biotin-H3K36me2 (28 to 48 residues) as substrate preincubated for 15 mins followed by substrate addition m... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific demethylase 4A (Homo sapiens (Human)) | BDBM50153093 (CHEMBL3775668) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 143 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research Curated by ChEMBL | Assay Description Inhibition of human N-terminal His-tagged KDM4A (1 to 359 residues) expressed in Escherichia coli using biotin-H3K9me3 as substrate preincubated for ... | J Med Chem 59: 1388-409 (2016) BindingDB Entry DOI: 10.7270/Q2W097TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||