 Found 6 hits for monomerid = 50158582
Found 6 hits for monomerid = 50158582 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50158582
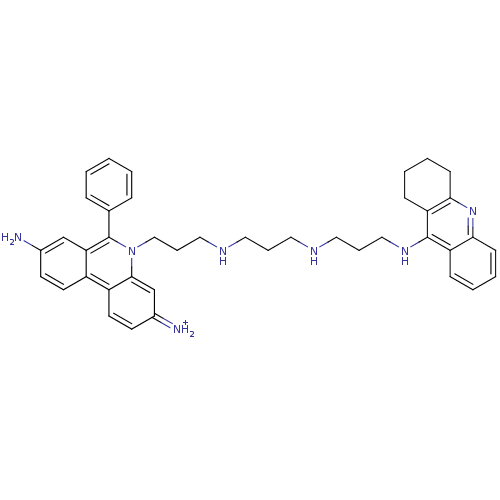
(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Competitive inhibition constant for Acetylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50158582

(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.55 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of Acetylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50158582

(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human AchE |
J Med Chem 51: 347-72 (2008)
Article DOI: 10.1021/jm7009364
BindingDB Entry DOI: 10.7270/Q25B039W |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50158582

(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63.7 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of Butyrylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50158582

(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.49 | n/a | n/a | n/a | n/a | n/a | n/a |
Sichuan University
Curated by ChEMBL
| Assay Description
Inhibition of AChE |
Eur J Med Chem 45: 1167-72 (2010)
Article DOI: 10.1016/j.ejmech.2009.12.038
BindingDB Entry DOI: 10.7270/Q25H7GFM |
More data for this
Ligand-Target Pair | |
Carboxylic ester hydrolase
(Equus caballus (Horse)) | BDBM50158582

(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibitory concentration against amyloid-beta aggregation |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data