

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50162959 (11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibenzo[e,h]azulen-2-ylmethyl)-dimethyl-amine::CHEMBL366164
SMILES: CN(C)C[C@H]1C[C@H]2[C@H](O1)c1cc(F)ccc1Sc1ccccc21
InChI Key: InChIKey=BSOLCASXROXPRZ-MUKKUYKPSA-N
Data: 10 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50162959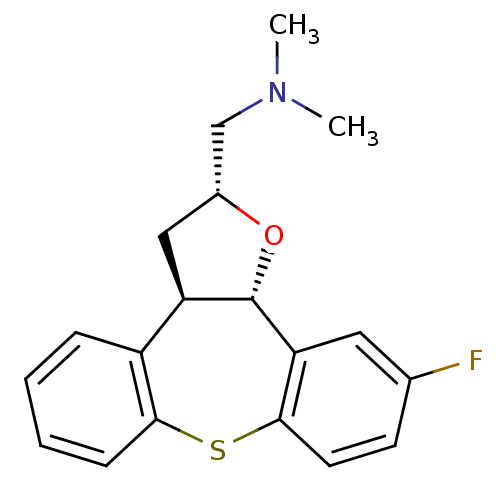 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-pyrilamine binding to human Histamine H1 receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2C (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-mesulergine binding to human 5-hydroxytryptamine 2C receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [125I]-R91150 binding to human 5-hydroxytryptamine 2A receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 7 (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-5-HT binding to human 5-hydroxytryptamine 7 receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-spiperone binding to human Dopamine receptor D2 | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [125I]-iodosulpride binding to human Dopamine receptor D3 | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(1A) dopamine receptor (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-SCH-23,390 binding to human Dopamine receptor D1 | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Norepinephrine Monoamine transporters (Rattus norvegicus) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | Reactome pathway KEGG UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-nisoxetine binding to rat Norepinephrine transpoter | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-2C adrenergic receptor (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-rauwolscine binding to Alpha-2C adrenergic receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-2A adrenergic receptor (Homo sapiens (Human)) | BDBM50162959 ((11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 156 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development Curated by ChEMBL | Assay Description Inhibition of [3H]-rauwolscine binding to Alpha-2A adrenergic receptor | J Med Chem 48: 1709-12 (2005) Article DOI: 10.1021/jm049632c BindingDB Entry DOI: 10.7270/Q2MC8ZHB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||