

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50172414 2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-ethoxy)-4-methyl-quinolin-6-yl]-acetamide::CHEMBL194837
SMILES: CN(C)CCOc1cc(C)c2cc(NC(=O)COc3ccc(Cl)cc3Cl)ccc2n1
InChI Key: InChIKey=GKDBTHSYQZTDAJ-UHFFFAOYSA-N
Data: 15 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50172414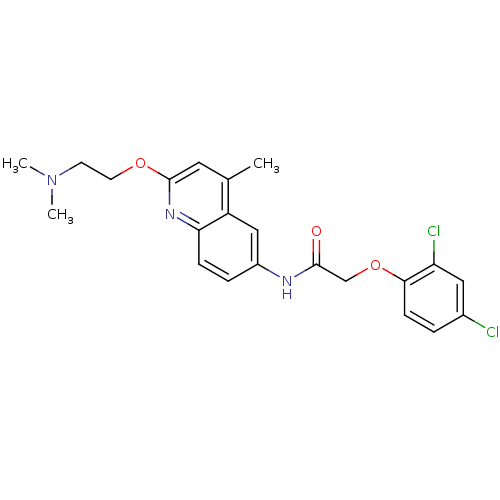 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against muscarinic acetylcholine receptor M1 | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| GPR145 (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 8.70 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Displacement of 15 pM [125I]-MCH from human MCH2R expressed in CHO-K1 in whole-cell binding assay | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1B (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against 5-hydroxytryptamine 1B receptor | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanin-concentrating hormone receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 1.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against 10 nM MCH-induced IP3 accumulation in CHO-K1 cells expressing human MCH1R after incubation with [3H]-myo-inositol | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanin-concentrating hormone receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 85 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Displacement of 15 pM [125I]-MCH from human MCH1R expressed in CHO-K1 in whole-cell binding assay | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2C (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against human 5-hydroxytryptamine 2C receptor expressed in CHO-K1 cells using [3H]-mesulergine | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against dopamine receptor D3 | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against histamine H1 receptor | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium-dependent dopamine transporter (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | >10 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against dopamine transporter | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(1A) dopamine receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against dopamine receptor D1 | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 0.610 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against 5-hydroxytryptamine 2A receptor | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against muscarinic acetylcholine receptor M2 | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2C (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against human 5-hydroxytryptamine 2C receptor expressed in CHO-K1 cells using [3H]-mesulergine | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanin-concentrating hormone receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Displacement of 150p M [125I]-MCH from human MCH1R expressed in CHO-K1 cells | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H2 Receptor (Homo sapiens (Human)) | BDBM50172414 (2-(2,4-Dichloro-phenoxy)-N-[2-(2-dimethylamino-eth...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 0.870 | n/a | n/a | n/a | n/a | n/a | n/a |
7TM Pharma A/S Curated by ChEMBL | Assay Description Inhibitory concentration against histamine H2 receptor | J Med Chem 48: 5684-97 (2005) Article DOI: 10.1021/jm050103y BindingDB Entry DOI: 10.7270/Q2H41QZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||