

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50182221 CHEMBL381366::N-((R)-6-((tert-butylamino)methyl)-1,2,3,4-tetrahydronaphthalen-1-yl)-2-((S)-1-(3-(trifluoromethyl)phenylsulfonyl)piperidin-2-yl)acetamide
SMILES: CC(C)(C)NCc1ccc2[C@@H](CCCc2c1)NC(=O)C[C@@H]1CCCCN1S(=O)(=O)c1cccc(c1)C(F)(F)F
InChI Key: InChIKey=BFWDXOBUPPVYOP-JYFHCDHNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221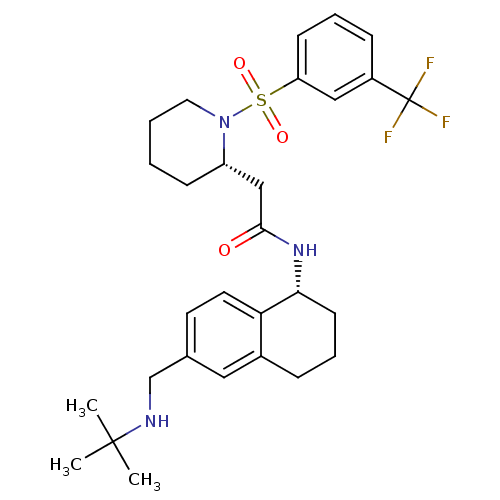 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc. Curated by ChEMBL | Assay Description Antagonist activity at human bradykinin B1 receptor | Bioorg Med Chem Lett 21: 3384-9 (2011) Article DOI: 10.1016/j.bmcl.2011.03.115 BindingDB Entry DOI: 10.7270/Q2FX79R6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Displacement of [3H]DAK from human bradykinin B1 receptor expressed in CHO-D-/aequorin cells by rapid filtration technique | Bioorg Med Chem Lett 18: 4477-81 (2008) Article DOI: 10.1016/j.bmcl.2008.07.055 BindingDB Entry DOI: 10.7270/Q2F76CCT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc. Curated by ChEMBL | Assay Description Antagonist activity at human bradykinin B1 receptor expressed in CHO cells assessed as inhibition of agonist-induced calcium efflux by aquerin based ... | Bioorg Med Chem Lett 21: 3384-9 (2011) Article DOI: 10.1016/j.bmcl.2011.03.115 BindingDB Entry DOI: 10.7270/Q2FX79R6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Inhibition of human B1 receptor | Bioorg Med Chem Lett 16: 2071-5 (2006) Article DOI: 10.1016/j.bmcl.2006.01.069 BindingDB Entry DOI: 10.7270/Q2SN08K7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Inhibition of human B1 receptor by calcium influx functional assay | Bioorg Med Chem Lett 16: 2071-5 (2006) Article DOI: 10.1016/j.bmcl.2006.01.069 BindingDB Entry DOI: 10.7270/Q2SN08K7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bradykinin B1 receptor (Homo sapiens (Human)) | BDBM50182221 (CHEMBL381366 | N-((R)-6-((tert-butylamino)methyl)-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Antagonist activity at human bradykinin B1 receptor expressed in CHO-D-/aequorin cells | Bioorg Med Chem Lett 18: 4477-81 (2008) Article DOI: 10.1016/j.bmcl.2008.07.055 BindingDB Entry DOI: 10.7270/Q2F76CCT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||