

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50183557 CHEMBL442483::H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-Nle-Trp-Ac
SMILES: CCCC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccccc1)NN[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@H](C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O)C(=O)N[C@@H](Cc1cn(C(C)=O)c2ccccc12)C(O)=O
InChI Key: InChIKey=RBNPCZIQGBMFDV-FPWNPRTMSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50183557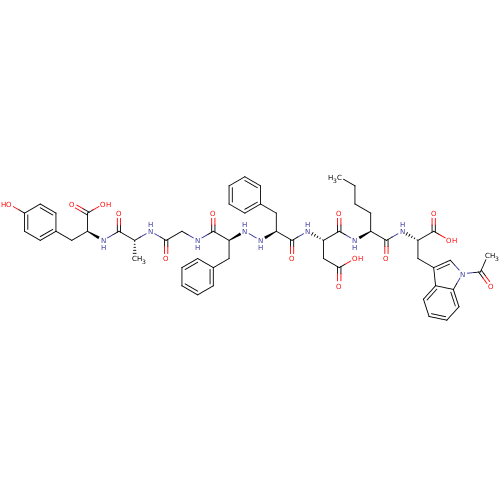 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in HN9.10 cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor (Homo sapiens (Human)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Displacement of [125I]CCK8 from human CCK2 receptor expressed in HEK293 cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 76 | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Inhibition of [35S]GTP-gamma-S binding to rat mu opioid receptor expressed in CHO cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 76 | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in HN9.10 cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor by inhibition of muscle contraction in electrically stimulated isolated guinea pig ileum | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor by inhibition of muscle contraction in electrically stimulated isolated mouse vas deferens | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor (Homo sapiens (Human)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Displacement of [125I]CCK8 from human CCK2 receptor expressed in HEK293 cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 55 | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Inhibition of [35S]GTP-gamma-S binding to human delta opioid receptor expressed in CHO cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50183557 (CHEMBL442483 | H-Tyr-D-Ala-Gly-Phe-NH-NH-Phe-Asp-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 62 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in HN9.10 cells | J Med Chem 49: 1773-80 (2006) Article DOI: 10.1021/jm050851n BindingDB Entry DOI: 10.7270/Q2FT8MVM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||