

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50192138 CHEMBL215904::methyl 2-(ethyl(4-(1,1,1,3,3,3-hexafluoro-2-hydroxypropan-2-yl)phenyl)amino)-2-phenylpropanoate
SMILES: CCN(c1ccc(cc1)C(O)(C(F)(F)F)C(F)(F)F)C(C)(C(=O)OC)c1ccccc1
InChI Key: InChIKey=GXMQHNQBWIJYMO-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50192138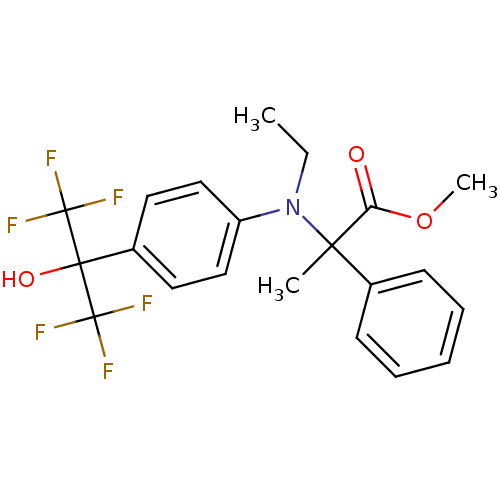 (CHEMBL215904 | methyl 2-(ethyl(4-(1,1,1,3,3,3-hexa...) | GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Transactivation of LXRbeta by luciferase reporter gene assay | Bioorg Med Chem Lett 16: 5231-7 (2006) Article DOI: 10.1016/j.bmcl.2006.06.081 BindingDB Entry DOI: 10.7270/Q2HD7V80 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-alpha (Homo sapiens (Human)) | BDBM50192138 (CHEMBL215904 | methyl 2-(ethyl(4-(1,1,1,3,3,3-hexa...) | GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Binding affinity to LXRalpha by radioligand displacement assay | Bioorg Med Chem Lett 16: 5231-7 (2006) Article DOI: 10.1016/j.bmcl.2006.06.081 BindingDB Entry DOI: 10.7270/Q2HD7V80 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50192138 (CHEMBL215904 | methyl 2-(ethyl(4-(1,1,1,3,3,3-hexa...) | GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Binding affinity to LXRbeta by radioligand displacement assay | Bioorg Med Chem Lett 16: 5231-7 (2006) Article DOI: 10.1016/j.bmcl.2006.06.081 BindingDB Entry DOI: 10.7270/Q2HD7V80 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-alpha (Homo sapiens (Human)) | BDBM50192138 (CHEMBL215904 | methyl 2-(ethyl(4-(1,1,1,3,3,3-hexa...) | GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Transactivation of LXRalpha by luciferase reporter gene assay | Bioorg Med Chem Lett 16: 5231-7 (2006) Article DOI: 10.1016/j.bmcl.2006.06.081 BindingDB Entry DOI: 10.7270/Q2HD7V80 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||