

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50210579 (2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benzyl-5-(2-carboxyethyl)-17-(carboxymethyl)-13-hydroxy-14,20-diisobutyl-8-isopropyl-11-methyl-4,7,10,16,19,22-hexaoxo-3,6,9,15,18,21-hexaazahexacosane-1,26-dioic acid::CHEMBL390456::OM00-3
SMILES: CC(C)C[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCC(O)=O)[C@@H](O)C[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(O)=O
InChI Key: InChIKey=CFIWWTUUSMMDDA-IQRKPKHNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50210579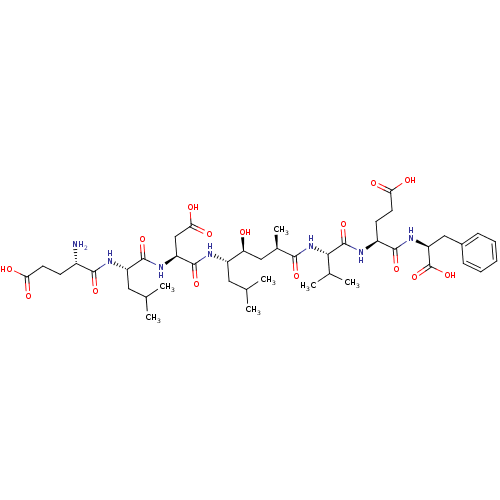 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | MMDB Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela Curated by ChEMBL | Assay Description Inhibition of human recombinant BACE1 ectodomain (1 to 460 amino acids) assessed as inhibition of proteolytic cleavage of Rhodamine-EVNLDAEFK-Quenche... | J Med Chem 54: 3081-5 (2011) Article DOI: 10.1021/jm101568y BindingDB Entry DOI: 10.7270/Q20C4W7T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50210579 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | MMDB Article PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Biologia Experimental e Tecnol£gica Curated by ChEMBL | Assay Description Inhibition of active BACE1 (unknown origin) | J Med Chem 58: 5408-18 (2015) Article DOI: 10.1021/acs.jmedchem.5b00658 BindingDB Entry DOI: 10.7270/Q2HX1FDQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50210579 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | MMDB Article PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Biologia Experimental e Tecnol£gica Curated by ChEMBL | Assay Description Inhibition of human recombinant BACE1 using Mca-SEVNLDAEFK-DNP substrate assessed as substrate hydrolysis after 2 hrs by HPLC-FLU analysis | J Med Chem 58: 5408-18 (2015) Article DOI: 10.1021/acs.jmedchem.5b00658 BindingDB Entry DOI: 10.7270/Q2HX1FDQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM50210579 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Biologia Experimental e Tecnol£gica Curated by ChEMBL | Assay Description Apparent inhibition of human recombinant BACE2 using Mca-SEVNLDAEFK-DNP substrate assessed as substrate hydrolysis after 1 hr by HPLC-FLU analysis | J Med Chem 58: 5408-18 (2015) Article DOI: 10.1021/acs.jmedchem.5b00658 BindingDB Entry DOI: 10.7270/Q2HX1FDQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50210579 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | MMDB Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Technische Universit£t M£nchen Curated by ChEMBL | Assay Description Inhibition of full-domain of BACE1 expressed in HEK393 cells | Bioorg Med Chem Lett 19: 4427-31 (2009) Article DOI: 10.1016/j.bmcl.2009.05.053 BindingDB Entry DOI: 10.7270/Q2RF5V22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50210579 ((2S,5S,8S,11R,13S,14S,17S,20S,23S)-23-amino-2-benz...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid UniChem Similars | MMDB Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Technische Universität München Curated by ChEMBL | Assay Description Inhibition of human BACE1 expressed in HEK293 cells | Bioorg Med Chem 15: 4136-43 (2007) Article DOI: 10.1016/j.bmc.2007.03.072 BindingDB Entry DOI: 10.7270/Q24M9478 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||