

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50237994 CHEMBL4082723
SMILES: NC(CO)(CO)CO.CCc1cc2oc(=O)n(CCC(O)=O)c2cc1Cl
InChI Key: InChIKey=ZHMLSGOGTNVURP-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kynurenine 3-monooxygenase (Homo sapiens (Human)) | BDBM50237994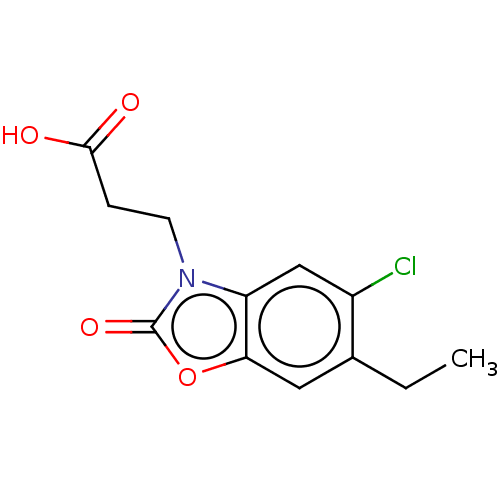 (CHEMBL4082723) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Compound was tested for binding affinity towards opioid receptor delta 1 by displacing [3H]DPDPE radioligand in rat brain P2 synaptosomes membranes. | Bioorg Med Chem Lett 27: 2023-2028 (2017) Article DOI: 10.1016/j.bmcl.2017.02.078 BindingDB Entry DOI: 10.7270/Q23R0W54 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine 3-monooxygenase (Homo sapiens (Human)) | BDBM50237994 (CHEMBL4082723) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 316 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of full length human KMO expressed in HEK293 cells using kynurenine as substrate measured after 20 hrs by LC-MS/MS analysis | J Med Chem 60: 3383-3404 (2017) Article DOI: 10.1021/acs.jmedchem.7b00055 BindingDB Entry DOI: 10.7270/Q26112SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine 3-monooxygenase (Homo sapiens (Human)) | BDBM50237994 (CHEMBL4082723) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of full length human GST-tagged KMO expressed in baculovirus infected Sf9 insect cell membranes using kynurenine as substrate measured aft... | J Med Chem 60: 3383-3404 (2017) Article DOI: 10.1021/acs.jmedchem.7b00055 BindingDB Entry DOI: 10.7270/Q26112SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine 3-monooxygenase (Homo sapiens (Human)) | BDBM50237994 (CHEMBL4082723) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 316 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of full length human KMO expressed in HEK293 cells using L-kynurenine as substrate measured after 20 hrs by LC-MS/MS method | Bioorg Med Chem Lett 27: 2023-2028 (2017) Article DOI: 10.1016/j.bmcl.2017.02.078 BindingDB Entry DOI: 10.7270/Q23R0W54 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||