

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50240419 (E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one::6-shogaol::CHEMBL25948::[(E)-1-(4-hydroxy-3-methoxyphenyl)-dec-4-en-3-one]::[6]-shogaol::trans-6-shogaol
SMILES: CCCCCC=CC(=O)CCc1ccc(O)c(OC)c1
InChI Key: InChIKey=OQWKEEOHDMUXEO-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 1A (Homo sapiens (Human)) | BDBM50240419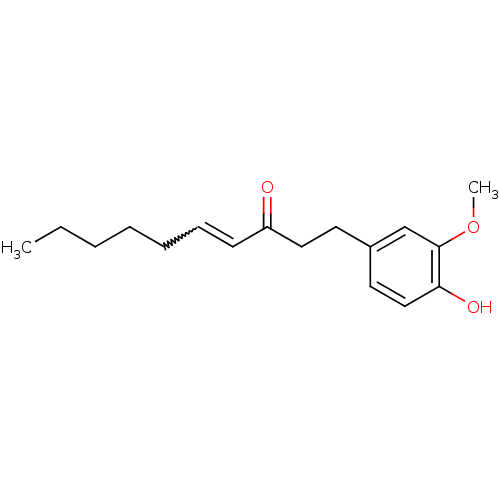 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry and Applied Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]8-OH-DPAT from 5HT1A receptor expressed in HEK293 cells after 2 hrs by liquid scintillation counting | Bioorg Med Chem 18: 3345-51 (2010) Article DOI: 10.1016/j.bmc.2010.02.062 BindingDB Entry DOI: 10.7270/Q2RN381W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily A member 1 (Rattus norvegicus) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza Universit£ di Roma Curated by ChEMBL | Assay Description Antagonist activity at rat TRPA1 ion channel expressed in HEK293 cells assessed as inhibition of AITC-induced calcium influx incubated for 5 mins pri... | Bioorg Med Chem Lett 22: 1674-7 (2012) Article DOI: 10.1016/j.bmcl.2011.12.113 BindingDB Entry DOI: 10.7270/Q20002JM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 320 | n/a | n/a | n/a | n/a |
Sapienza Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at human TRPV1 ion channel expressed in HEK293 cells assessed as calcium influx by fluo-4-Am-based fluorimetry | Bioorg Med Chem Lett 22: 1674-7 (2012) Article DOI: 10.1016/j.bmcl.2011.12.113 BindingDB Entry DOI: 10.7270/Q20002JM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.77E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Toyama Medical and Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human CYP3A4 by radiometric assay | J Nat Prod 67: 1079-83 (2004) Article DOI: 10.1021/np030556a BindingDB Entry DOI: 10.7270/Q2668CZP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Leukotriene A4 hydrolase (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.46E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mansoura University Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human LTA4H epoxide hydrolase activity expressed in Escherichia coli assessed as reduction in LTB4 pr... | Bioorg Med Chem 25: 1277-1285 (2017) Article DOI: 10.1016/j.bmc.2016.12.048 BindingDB Entry DOI: 10.7270/Q2SJ1NMK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Advanced Industrial Science and Technology (AIST) Curated by ChEMBL | Assay Description Inhibition of 5LOX | Bioorg Med Chem 16: 10332-7 (2008) Article DOI: 10.1016/j.bmc.2008.10.038 BindingDB Entry DOI: 10.7270/Q24M94C2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily A member 1 (Rattus norvegicus) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a |
Sapienza Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at rat TRPA1 ion channel expressed in HEK293 cells assessed as calcium influx by fluo-4-Am-based fluorimetry | Bioorg Med Chem Lett 22: 1674-7 (2012) Article DOI: 10.1016/j.bmcl.2011.12.113 BindingDB Entry DOI: 10.7270/Q20002JM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Leukotriene A4 hydrolase (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mansoura University Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human LTA4H epoxide hydrolase activity expressed in Escherichia coli assessed as reduction in LTB4 pr... | Bioorg Med Chem 25: 1277-1285 (2017) Article DOI: 10.1016/j.bmc.2016.12.048 BindingDB Entry DOI: 10.7270/Q2SJ1NMK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza Universit£ di Roma Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 ion channel expressed in HEK293 cells assessed as inhibition of capsaicin-induced calcium influx incubated for 5 m... | Bioorg Med Chem Lett 22: 1674-7 (2012) Article DOI: 10.1016/j.bmcl.2011.12.113 BindingDB Entry DOI: 10.7270/Q20002JM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50240419 ((E)-1-(4-Hydroxy-3-methoxy-phenyl)-dec-4-en-3-one ...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Toyama Medical and Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human CYP2D6 by radiometric assay | J Nat Prod 67: 1079-83 (2004) Article DOI: 10.1021/np030556a BindingDB Entry DOI: 10.7270/Q2668CZP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||