

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50255908 4-(5-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(5-fluoro-6-methylpyridin-2-yl)-1H-imidazol-2-yl)morpholine::CHEMBL520104
SMILES: Cc1nc(ccc1F)-c1nc([nH]c1-c1ccc2ncnn2c1)N1CCOCC1
InChI Key: InChIKey=CIRCIOIQLMLPCO-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TGF-beta receptor type-1 (Homo sapiens (Human)) | BDBM50255908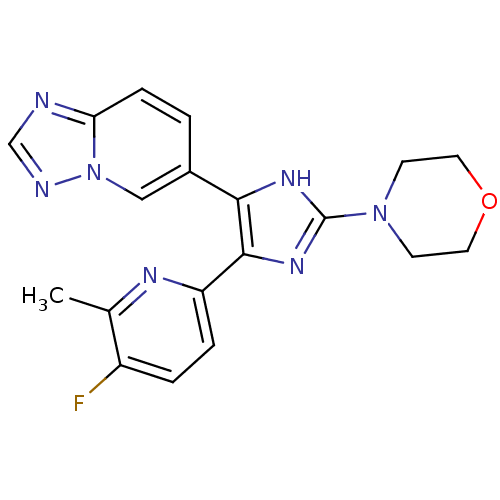 (4-(5-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(5-flu...) | PDB MMDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbot Laboratories Curated by ChEMBL | Assay Description Binding affinity to human TGFBR1 | Bioorg Med Chem Lett 19: 912-6 (2009) Article DOI: 10.1016/j.bmcl.2008.11.119 BindingDB Entry DOI: 10.7270/Q2FX799C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| TGF-beta receptor type-1 (Homo sapiens (Human)) | BDBM50255908 (4-(5-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(5-flu...) | PDB MMDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 513 | n/a | n/a | n/a | n/a |
Abbot Laboratories Curated by ChEMBL | Assay Description Inhibition of TGFBR1 (unknown origin) transfected in human HepG2 cells after 24 hrs by plasminogen activator inhibitor-luciferase reporter gene assay | Bioorg Med Chem Lett 19: 912-6 (2009) Article DOI: 10.1016/j.bmcl.2008.11.119 BindingDB Entry DOI: 10.7270/Q2FX799C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| TGF-beta receptor type-1 (Homo sapiens (Human)) | BDBM50255908 (4-(5-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(5-flu...) | PDB MMDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.63E+4 | n/a | n/a | n/a | n/a |
Abbot Laboratories Curated by ChEMBL | Assay Description Inhibition of p38alpha (unknown origin) | Bioorg Med Chem Lett 19: 912-6 (2009) Article DOI: 10.1016/j.bmcl.2008.11.119 BindingDB Entry DOI: 10.7270/Q2FX799C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||