

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50262420 7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl)-heptan-1-one::CHEMBL477372
SMILES: O=C(CCCCCCc1ccccc1)c1nnc(s1)-c1ccccn1
InChI Key: InChIKey=PVCCIDZCPLBVCC-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fatty-acid amide hydrolase 1 (aa 30-579) (Rattus norvegicus (rat)) | BDBM50262420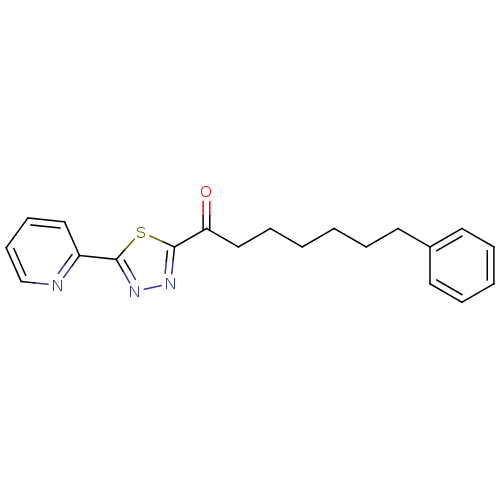 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of rat FAAH expressed in Escherichia coli | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (aa 30-579) (Rattus norvegicus (rat)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | 9.0 | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of rat FAAH expressed in Escherichia coli at pH 9.0 | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of human recombinant FAAH expressed in african green monkey COS7 cells | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (aa 30-579) (Rattus norvegicus (rat)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of rat FAAH expressed in Escherichia coli | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acyl-CoA: cholesterol acyltransferase (ACAT) (Homo sapiens (Human)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of TGH | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sn1-specific diacylglycerol lipase alpha (Homo sapiens (Human)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Leiden University Curated by ChEMBL | Assay Description Inhibition of full-length human DAGLalpha expressed in HEK293T cell membranes using para-nitrophenylbutyrate by colorimetric assay | J Med Chem 58: 9742-53 (2015) Article DOI: 10.1021/acs.jmedchem.5b01627 BindingDB Entry DOI: 10.7270/Q2D50QZ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutral cholesterol ester hydrolase 1 (Homo sapiens (Human)) | BDBM50262420 (7-Phenyl-1-(5-(pyridin-2-yl)-1,3,4-thiadiazol-2-yl...) | KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Chemical Biology Curated by ChEMBL | Assay Description Inhibition of KIAA1363 hydrolase | J Med Chem 51: 4392-403 (2008) Article DOI: 10.1021/jm800136b BindingDB Entry DOI: 10.7270/Q26D5TW3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||