

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50275889 CHEMBL508102::carbimazole
SMILES: CCOC(=O)n1ccn(C)c1=S
InChI Key: InChIKey=CFOYWRHIYXMDOT-UHFFFAOYSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bile salt export pump (Homo sapiens (Human)) | BDBM50275889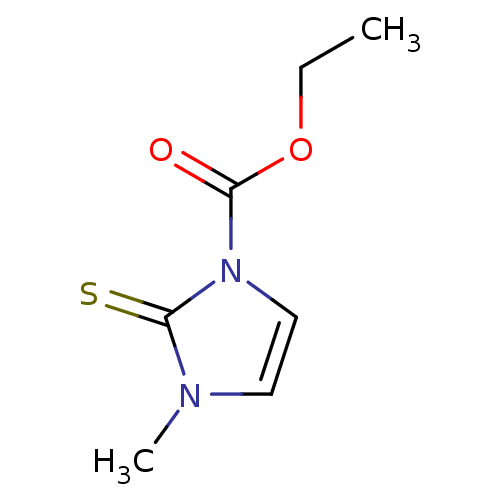 (CHEMBL508102 | carbimazole) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Ability to inhibit HMG-CoA reductase (HMGR) by CoA reductase inhibition screen (COR) in rats | Drug Metab Dispos 40: 2332-41 (2012) Article DOI: 10.1124/dmd.112.047068 BindingDB Entry DOI: 10.7270/Q2ZP488M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lactoperoxidase (Homo sapiens (Human)) | BDBM50275889 (CHEMBL508102 | carbimazole) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.04E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Science Curated by ChEMBL | Assay Description Inhibition of lactoperoxidase (unknown origin)-catalyzed iodination of L-tyrosine assessed as 3,5-diiodo-L-tyrosine formation by HPLC | J Med Chem 51: 7313-7 (2009) Article DOI: 10.1021/jm800894m BindingDB Entry DOI: 10.7270/Q2Z31ZGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||