

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50277668 (R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-dihydro-6H-pyrido[3,4-b]pyrrolizin-8-yl)acetic acid::CHEMBL483991
SMILES: CC(C)c1nccc2n3CC[C@H](CC(O)=O)c3c(Sc3ccc(Cl)c(Cl)c3)c12
InChI Key: InChIKey=LUZBJNWFEDHIPR-GFCCVEGCSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prostanoid DP receptor (Homo sapiens (Human)) | BDBM50277668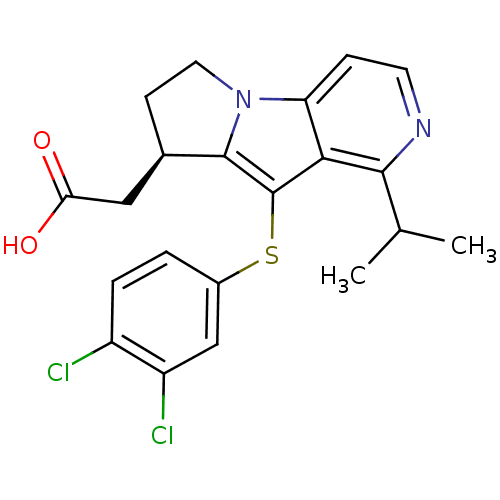 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Displacement of radioligand from human recombinant DP1 receptor expressed in HEK293 cells by scintillation proximity assay | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 851 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to EP3 receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostanoid TP receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 935 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Displacement of radioligand from human recombinant TP receptor expressed in HEK293 cells by scintillation proximity assay | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP2 subtype (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to EP2 receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| G protein-coupled receptor 44 (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.79E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to CRTH2 receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP4 subtype (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | >1.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to EP4 receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin F2-alpha receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | >2.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to FP receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostanoid IP receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | >2.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to IP receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (EP1) (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.85E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Binding affinity to EP1 receptor | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostanoid TP receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Antagonist activity at TP1 in human platelet-rich plasma assessed as inhibition of U44619-induced platelet aggregation | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostanoid DP receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Antagonist activity at DP1 in human platelet-rich plasma assessed as inhibition of PGD2-induced [125I]cAMP production preincubated 10 mins before PGD... | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostanoid DP receptor (Homo sapiens (Human)) | BDBM50277668 ((R)-2-(9-(3,4-dichlorophenylthio)-1-isopropyl-7,8-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Antagonist activity at DP1 in washed human platelet assessed as inhibition of PGD2-induced [125I]cAMP production preincubated 10 mins before PGD2 cha... | Bioorg Med Chem Lett 19: 2125-8 (2009) Article DOI: 10.1016/j.bmcl.2009.03.010 BindingDB Entry DOI: 10.7270/Q2W9593R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||