

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50277753 2-[3-(7-Hydroxy-benzo[d]imidazo[2,1-b]thiazol-2-yl)-propionylamino]-cyclohex-1-enecarboxylic acid::CHEMBL520513
SMILES: OC(=O)C1=C(CCCC1)NC(=O)CCc1cn2c(n1)sc1cc(O)ccc21
InChI Key: InChIKey=GGGKZUYCIJIKSM-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50277753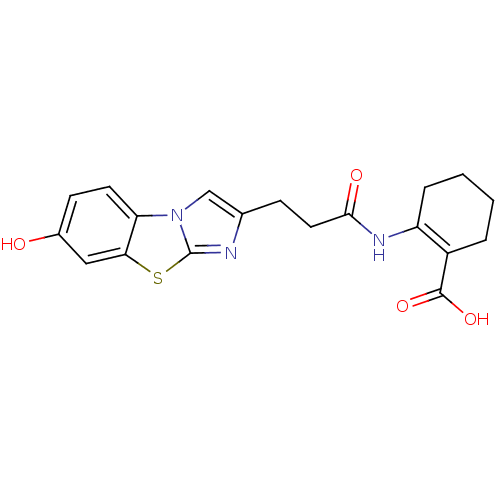 (2-[3-(7-Hydroxy-benzo[d]imidazo[2,1-b]thiazol-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes using diclofenac substrate | J Med Chem 52: 2587-602 (2009) Article DOI: 10.1021/jm900151e BindingDB Entry DOI: 10.7270/Q2RJ4JDG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hydroxycarboxylic acid receptor 2 (Homo sapiens (Human)) | BDBM50277753 (2-[3-(7-Hydroxy-benzo[d]imidazo[2,1-b]thiazol-2-yl...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 76 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at human GPR109A expressed in CHOK1 cells by [35S]GTPgammaS binding assay | J Med Chem 52: 2587-602 (2009) Article DOI: 10.1021/jm900151e BindingDB Entry DOI: 10.7270/Q2RJ4JDG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hydroxycarboxylic acid receptor 2 (Homo sapiens (Human)) | BDBM50277753 (2-[3-(7-Hydroxy-benzo[d]imidazo[2,1-b]thiazol-2-yl...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]niacin from human GPR109A expressed in CHO cells | J Med Chem 52: 2587-602 (2009) Article DOI: 10.1021/jm900151e BindingDB Entry DOI: 10.7270/Q2RJ4JDG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||