

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50283765 CHEMBL321313::N-{3-[3-Butyl-4-(3-fluoro-2'-(ethyloxtycarbonyl)sulfamoyl-5'-propyl-biphenyl-4-ylmethyl)-5-oxo-4,5-dihydro-[1,2,4]triazol-1-yl]-4-chloro-phenyl}-acetamide
SMILES: CCCCc1nn(-c2cc(NC(C)=O)ccc2Cl)c(=O)n1Cc1ccc(cc1F)-c1cc(CCC)ccc1S(=O)(=O)NC(=O)OCC
InChI Key: InChIKey=AVPYNETYRCMACW-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiotensin II type 1a (AT-1a) receptor (RABBIT) | BDBM50283765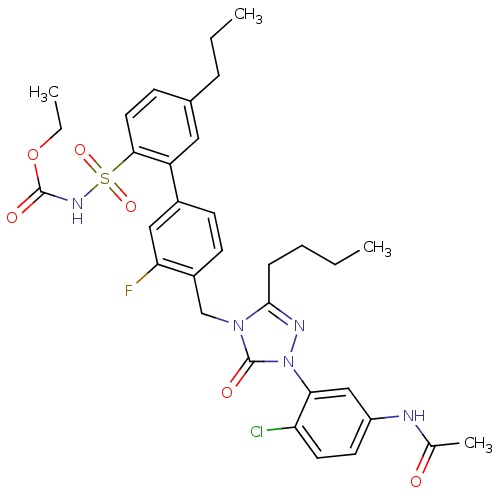 (CHEMBL321313 | N-{3-[3-Butyl-4-(3-fluoro-2'-(ethyl...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro ability of the compound to inhibit the binding of radioligand 125I[Sar1,IIe8]AII to AT1 receptor from rabbit aorta | Bioorg Med Chem Lett 4: 2787-2792 (1994) Article DOI: 10.1016/S0960-894X(01)80595-3 BindingDB Entry DOI: 10.7270/Q2WS8T68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin II receptor (Homo sapiens (Human)) | BDBM50283765 (CHEMBL321313 | N-{3-[3-Butyl-4-(3-fluoro-2'-(ethyl...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro inhibitory concentration of the compound against AT2 receptor from human adrenal tissues. | Bioorg Med Chem Lett 4: 2787-2792 (1994) Article DOI: 10.1016/S0960-894X(01)80595-3 BindingDB Entry DOI: 10.7270/Q2WS8T68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin II receptor (Homo sapiens (Human)) | BDBM50283765 (CHEMBL321313 | N-{3-[3-Butyl-4-(3-fluoro-2'-(ethyl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro inhibitory concentration of the compound against AT1 receptor from human adrenal tissues. | Bioorg Med Chem Lett 4: 2787-2792 (1994) Article DOI: 10.1016/S0960-894X(01)80595-3 BindingDB Entry DOI: 10.7270/Q2WS8T68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin II AT2 (RAT) | BDBM50283765 (CHEMBL321313 | N-{3-[3-Butyl-4-(3-fluoro-2'-(ethyl...) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro ability of the compound to inhibit the binding of radioligand 125I[Sar1,IIe8]AII to AT2 receptor from rat midbrain | Bioorg Med Chem Lett 4: 2787-2792 (1994) Article DOI: 10.1016/S0960-894X(01)80595-3 BindingDB Entry DOI: 10.7270/Q2WS8T68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||