

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50288939 CHEMBL345187::Quinoline-2-carboxylic acid {(R)-1-[(1S,2R)-1-benzyl-3-((3S,4aS,8aS)-3-tert-butylcarbamoyl-octahydro-isoquinolin-2-yl)-2-hydroxy-propylcarbamoyl]-2-methanesulfonyl-2-methyl-propyl}-amide
SMILES: CC(C)(C)NC(=O)[C@@H]1C[C@@H]2CCCC[C@@H]2CN1C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)c1ccc2ccccc2n1)C(C)(C)S(C)(=O)=O
InChI Key: InChIKey=QJMAYXGAURCBTD-GIKUREFASA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM50288939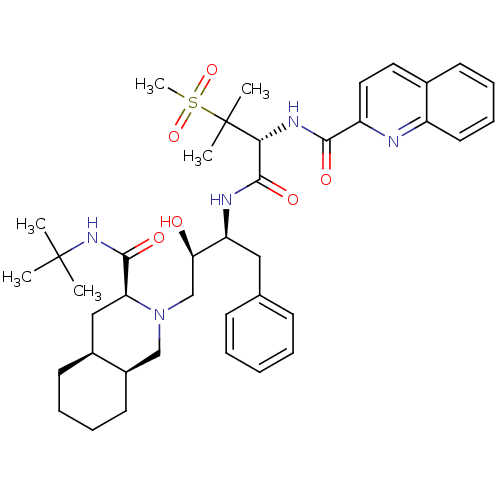 (CHEMBL345187 | Quinoline-2-carboxylic acid {(R)-1-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | 0.172 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against P2 site in HIV protease. | Bioorg Med Chem Lett 6: 585-588 (1996) Article DOI: 10.1016/0960-894X(96)00086-8 BindingDB Entry DOI: 10.7270/Q2KH0N9Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||