

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50301391 1-[4-(4-methylphenyl)butyl]-alpha,alpha-diphenyl-4-piperdine-methanol::CHEMBL571389
SMILES: Cc1ccc(CCCCN2CCC(CC2)C(O)(c2ccccc2)c2ccccc2)cc1
InChI Key: InChIKey=VFCNPRIWDSWJIG-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50301391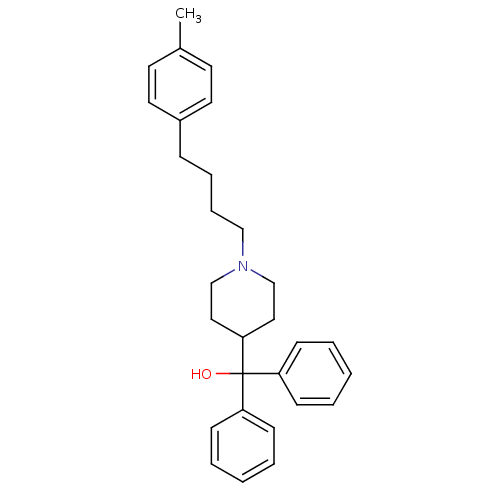 (1-[4-(4-methylphenyl)butyl]-alpha,alpha-diphenyl-4...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Schering Plough Research Institute Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human recombinant histamine H1 receptor expressed in CHO cell by betaplate scintillation counting | Bioorg Med Chem Lett 19: 5043-7 (2009) Article DOI: 10.1016/j.bmcl.2009.07.047 BindingDB Entry DOI: 10.7270/Q2NP24HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50301391 (1-[4-(4-methylphenyl)butyl]-alpha,alpha-diphenyl-4...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 396 | n/a | n/a | n/a | n/a | n/a | n/a |
The Schering Plough Research Institute Curated by ChEMBL | Assay Description Inhibition of human ERG in L929 cells by whole cell patch-clamp assay | Bioorg Med Chem Lett 19: 5043-7 (2009) Article DOI: 10.1016/j.bmcl.2009.07.047 BindingDB Entry DOI: 10.7270/Q2NP24HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50301391 (1-[4-(4-methylphenyl)butyl]-alpha,alpha-diphenyl-4...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 396 | n/a | n/a | n/a | n/a | n/a | n/a |
The Schering Plough Research Institute Curated by ChEMBL | Assay Description Inhibition of human ERG in L929 cells at 10 uM by whole cell patch-clamp assay | Bioorg Med Chem Lett 19: 5043-7 (2009) Article DOI: 10.1016/j.bmcl.2009.07.047 BindingDB Entry DOI: 10.7270/Q2NP24HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||