

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50303955 (E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25-dienyl)-2-nonylpropenoic Acid::CHEMBL578390::E3330::US9624235, Compound E3330
SMILES: CCCCCCCCCC(=CC1=C(C)C(=O)C(OC)=C(OC)C1=O)C(O)=O
InChI Key: InChIKey=AALSSIXXBDPENJ-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DNA-(apurinic or apyrimidinic site) endonuclease (Homo sapiens (Human)) | BDBM50303955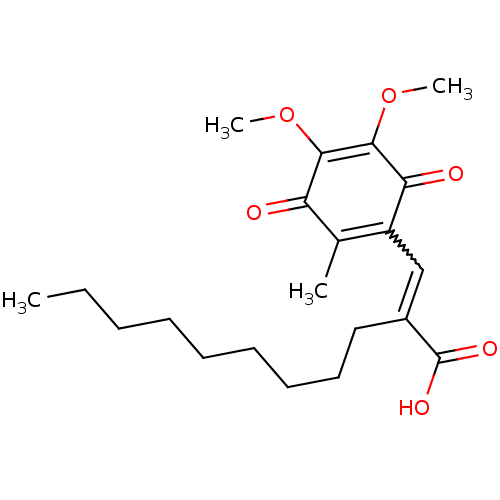 ((E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Indiana University School of Medicine, Indianapolis, IN 46202, USA | Assay Description E3330 or E3330-amide was preincubated with 2 μL of APE1 (reduced with 1.0 mM DTT for 10 min and then diluted to a concentration of 0.06 mM with ... | Biochemistry 52: 2955-66 (2013) Article DOI: 10.1021/bi400179m BindingDB Entry DOI: 10.7270/Q2TQ605T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA-(apurinic or apyrimidinic site) endonuclease (Homo sapiens (Human)) | BDBM50303955 ((E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of Ape1/ref-1 redox activity in presence of 0.02 mM DTT and human Hey-C2 cells nuclear extracts by EMSA | J Med Chem 53: 1200-10 (2010) Article DOI: 10.1021/jm9014857 BindingDB Entry DOI: 10.7270/Q2HD7VR8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA-(apurinic or apyrimidinic site) endonuclease (Homo sapiens (Human)) | BDBM50303955 ((E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Russian Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of redox activity of full length N-terminal hexa-His SUMO-fused human APE1 using HEX-labeled THF oligonucleotide as substrate preincubated... | Bioorg Med Chem 25: 2531-2544 (2017) Article DOI: 10.1016/j.bmc.2017.01.028 BindingDB Entry DOI: 10.7270/Q2P55R4B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA-(apurinic or apyrimidinic site) endonuclease (Homo sapiens (Human)) | BDBM50303955 ((E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a |
Russian Academy of Sciences Curated by ChEMBL | Assay Description Binding affinity to APE1 (unknown origin) by SPR assay | Bioorg Med Chem 25: 2531-2544 (2017) Article DOI: 10.1016/j.bmc.2017.01.028 BindingDB Entry DOI: 10.7270/Q2P55R4B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA-(apurinic or apyrimidinic site) endonuclease (Homo sapiens (Human)) | BDBM50303955 ((E)-3-(5,6-Dimethoxy-3-methyl-14-dioxocyclohexa-25...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.47E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pittsburgh—Of the Commonwealth System of Higher Education US Patent | Assay Description The initial screen was designed to look at the effect of potential inhibitors on APE1 repair kinetics. The approach can be used as a high throughput ... | US Patent US9624235 (2017) BindingDB Entry DOI: 10.7270/Q2SQ92GH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||