

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50317208 CHEMBL1088115::N-(adamantan-2-yl)-2-[cyclohexyl(ethyl)amino]acetamide
SMILES: CCN(CC(=O)NC1C2CC3CC(C2)CC1C3)C1CCCCC1
InChI Key: InChIKey=BGFPMHHVINUOLA-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 11-beta-hydroxysteroid dehydrogenase 1 (Homo sapiens (Human)) | BDBM50317208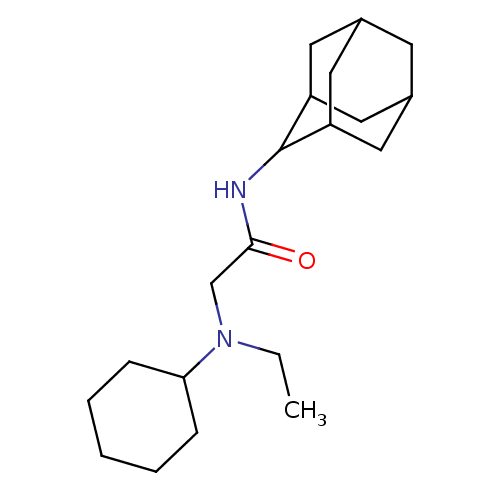 (CHEMBL1088115 | N-(adamantan-2-yl)-2-[cyclohexyl(e...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research& Development Curated by ChEMBL | Assay Description Inhibition of human 11-beta-HSD1 | Bioorg Med Chem Lett 20: 2897-902 (2010) Article DOI: 10.1016/j.bmcl.2010.03.032 BindingDB Entry DOI: 10.7270/Q2NV9JDJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 11-beta-hydroxysteroid dehydrogenase 1 (Homo sapiens (Human)) | BDBM50317208 (CHEMBL1088115 | N-(adamantan-2-yl)-2-[cyclohexyl(e...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research& Development Curated by ChEMBL | Assay Description Inhibition of human 11-beta-HSD1 expressed in HEK293 cells co-transfected with GRE-luciferase after 6 hrs by luciferase reporter gene assay | Bioorg Med Chem Lett 20: 2897-902 (2010) Article DOI: 10.1016/j.bmcl.2010.03.032 BindingDB Entry DOI: 10.7270/Q2NV9JDJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||