

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50322368 (S)-5-((S)-1-((S)-2-((S)-1-((S)-1-((S)-1-carboxy-3-methylbutylamino)-3,3-dimethyl-1-oxobutan-2-ylamino)-3-(4-hydroxyphenyl)-1-oxopropan-2-ylcarbamoyl)pyrrolidin-1-yl)-5-guanidino-1-oxopentan-2-ylamino)-N,N,N,4-tetramethyl-5-oxopentan-1-aminium::CHEMBL1172376
SMILES: CC(C)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](C)CCC[N+](C)(C)C)C(C)(C)C)C(O)=O
InChI Key: InChIKey=NWPZHWJQSGWXSJ-BJAKLJBYSA-O
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neurotensin receptor (Homo sapiens (Human)) | BDBM50322368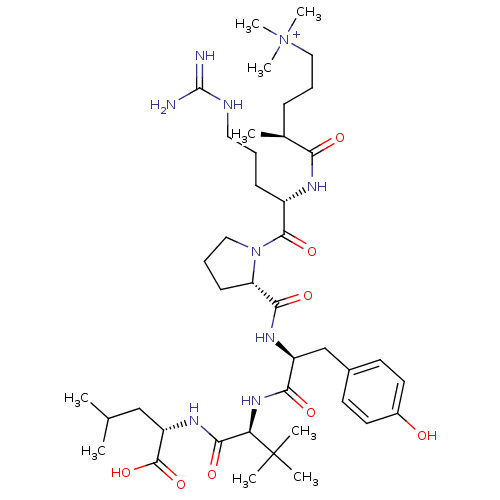 ((S)-5-((S)-1-((S)-2-((S)-1-((S)-1-((S)-1-carboxy-3...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Campus Curated by ChEMBL | Assay Description Binding affinity to human NTR2 by gamma counting | J Med Chem 53: 4623-32 (2010) Article DOI: 10.1021/jm100092s BindingDB Entry DOI: 10.7270/Q2DF6RDK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor 2 (Rattus norvegicus) | BDBM50322368 ((S)-5-((S)-1-((S)-2-((S)-1-((S)-1-((S)-1-carboxy-3...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Campus Curated by ChEMBL | Assay Description Displacement of [125I]NT from rat NTR2 by gamma counting | J Med Chem 53: 4623-32 (2010) Article DOI: 10.1021/jm100092s BindingDB Entry DOI: 10.7270/Q2DF6RDK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dopamine receptor D2L/neurotensin receptor NTS1 (Homo sapiens (Human)) | BDBM50322368 ((S)-5-((S)-1-((S)-2-((S)-1-((S)-1-((S)-1-carboxy-3...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Campus Curated by ChEMBL | Assay Description Binding affinity to human NTR1 by gamma counting | J Med Chem 53: 4623-32 (2010) Article DOI: 10.1021/jm100092s BindingDB Entry DOI: 10.7270/Q2DF6RDK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor 1 (Rattus norvegicus) | BDBM50322368 ((S)-5-((S)-1-((S)-2-((S)-1-((S)-1-((S)-1-carboxy-3...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 28 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Campus Curated by ChEMBL | Assay Description Agonist activity at rat NTR1 expressed in LTK cells assessed as calcium mobilization | J Med Chem 53: 4623-32 (2010) Article DOI: 10.1021/jm100092s BindingDB Entry DOI: 10.7270/Q2DF6RDK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||