

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: Cc1ccc(CN2[C@H]3CC[C@@H]2C[C@H](C3)Oc2cccc(c2)C(N)=O)cc1
InChI Key: InChIKey=NBCIMSDAMDKCQO-CEQLMSFQSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50327248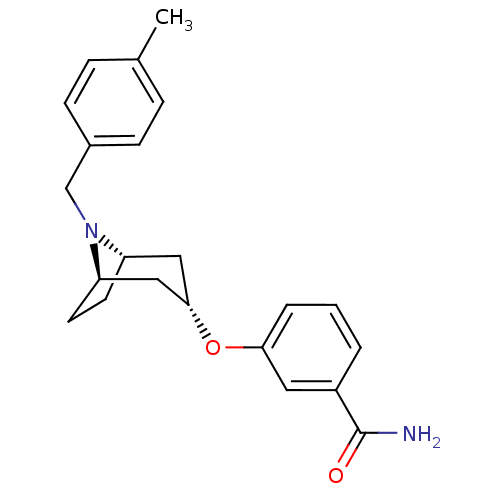 (CHEMBL1257821 | endo-3-(8-(4-methylbenzyl)-8-azabi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.09E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human delta opioid receptor assessed as inhibition of SNC80-stimulated [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 5847-52 (2010) Article DOI: 10.1016/j.bmcl.2010.07.113 BindingDB Entry DOI: 10.7270/Q23F4PV1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50327248 (CHEMBL1257821 | endo-3-(8-(4-methylbenzyl)-8-azabi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of human ERG | Bioorg Med Chem Lett 20: 5847-52 (2010) Article DOI: 10.1016/j.bmcl.2010.07.113 BindingDB Entry DOI: 10.7270/Q23F4PV1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50327248 (CHEMBL1257821 | endo-3-(8-(4-methylbenzyl)-8-azabi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human kappa opioid receptor assessed as inhibition of dynorphin A-induced [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 5847-52 (2010) Article DOI: 10.1016/j.bmcl.2010.07.113 BindingDB Entry DOI: 10.7270/Q23F4PV1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50327248 (CHEMBL1257821 | endo-3-(8-(4-methylbenzyl)-8-azabi...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 482 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human mu opioid receptor assessed as inhibition of DAMGO-stimulated [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 5847-52 (2010) Article DOI: 10.1016/j.bmcl.2010.07.113 BindingDB Entry DOI: 10.7270/Q23F4PV1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||