

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: C[C@H]1O[C@H](O[C@@H]2[C@@H](CO)O[C@H](O[C@@H]3[C@@H](CO)O[C@@H](O)[C@H](O)[C@H]3O)[C@H](O)[C@H]2O)[C@H](O)[C@@H](O)[C@@H]1N[C@H]1C=C(CO)[C@@H](O)[C@H](O)[C@H]1O
InChI Key: InChIKey=XUFXOAAUWZOOIT-SXARVLRPSA-N
PDB links: 2 PDB IDs match this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pancreatic alpha-amylase (Sus scrofa (Pig)) | BDBM50333465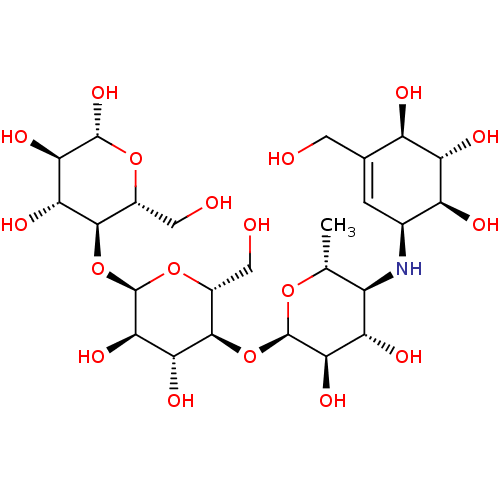 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
North Maharashtra University Curated by ChEMBL | Assay Description Inhibition of porcine pancreatic alpha amylase using starch as substrate by Bernfeld method | Bioorg Med Chem Lett 22: 7011-4 (2012) Article DOI: 10.1016/j.bmcl.2012.10.025 BindingDB Entry DOI: 10.7270/Q2FN17CD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Mus musculus (Mouse)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
North Maharashtra University Curated by ChEMBL | Assay Description Inhibition of liver glucosidase in Swiss mouse liver extracts | Bioorg Med Chem Lett 22: 7011-4 (2012) Article DOI: 10.1016/j.bmcl.2012.10.025 BindingDB Entry DOI: 10.7270/Q2FN17CD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sucrase-isomaltase, intestinal (Rattus norvegicus (Rat)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Kinki University Curated by ChEMBL | Assay Description Inhibition of rat intestinal sucrase | Bioorg Med Chem 19: 2015-22 (2011) Article DOI: 10.1016/j.bmc.2011.01.052 BindingDB Entry DOI: 10.7270/Q2J1044C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sucrase-isomaltase, intestinal (Rattus norvegicus (Rat)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.55E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Kinki University Curated by ChEMBL | Assay Description Inhibition of rat intestinal isomaltase | Bioorg Med Chem 19: 2015-22 (2011) Article DOI: 10.1016/j.bmc.2011.01.052 BindingDB Entry DOI: 10.7270/Q2J1044C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Kinki University Curated by ChEMBL | Assay Description Inhibition of rat intestinal maltase | Bioorg Med Chem 19: 2015-22 (2011) Article DOI: 10.1016/j.bmc.2011.01.052 BindingDB Entry DOI: 10.7270/Q2J1044C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article | n/a | n/a | 6.38E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of rat intestinal alpha-glucosidase using p-nitrophenyl- alpha-D-glucopyranoside as substrate preincubated for 5 min before addition of su... | Citation and Details Article DOI: 10.1007/s00044-010-9377-3 BindingDB Entry DOI: 10.7270/Q2S75K6S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL62 (Saccharomyces cerevisiae) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article | n/a | n/a | 9.08E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate preincubated for 10 min before substrate addition and ... | Citation and Details Article DOI: 10.1007/s00044-011-9699-9 BindingDB Entry DOI: 10.7270/Q2930X2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Homo sapiens (Human)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB NCI pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article | n/a | n/a | 1.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of alpha-glucosidase (unknown origin) using para-nitrophenyl-alpha-D-glucopyranoside as substrate incubated for 15 min prior to substrate ... | Citation and Details Article DOI: 10.1007/s00044-012-9974-4 BindingDB Entry DOI: 10.7270/Q22B91W5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sucrase-isomaltase, intestinal (Rattus norvegicus (Rat)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Xi'an Jiaotong University Curated by ChEMBL | Assay Description Inhibition of rat intestinal sucrase using sucrose as substrate assessed as D-glucose release from substrate preincubated for 15 mins measured after ... | Bioorg Med Chem 21: 5442-50 (2013) Article DOI: 10.1016/j.bmc.2013.06.002 BindingDB Entry DOI: 10.7270/Q2HD7X24 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article | n/a | n/a | 1.86E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of rat intestinal alpha-glucosidase using para-nitrophenyl-alpha-D-glucopyranoside as substrate incubated for 5 min prior to substrate add... | Citation and Details Article DOI: 10.1007/s00044-011-9583-7 BindingDB Entry DOI: 10.7270/Q25Q501R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Xi'an Jiaotong University Curated by ChEMBL | Assay Description Inhibition of rat intestinal maltase using maltose as substrate assessed as D-glucose release from substrate preincubated for 15 mins measured after ... | Bioorg Med Chem 21: 5442-50 (2013) Article DOI: 10.1016/j.bmc.2013.06.002 BindingDB Entry DOI: 10.7270/Q2HD7X24 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oligo-1,6-glucosidase IMA1 (Saccharomyces cerevisiae S288c (Baker's yeast)) | BDBM50333465 ((2R,3R,4R,5R,6R)-5-((2R,3R,4R,5S,6R)-5-((2R,3R,4S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 2.35E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Xi'an Jiaotong University Curated by ChEMBL | Assay Description Inhibition of Saccharomyces cerevisiae alpha-glucosidase assessed as 4-nitrophenol release from 4-nitrophenyl alpha-D-glucopyranoside preincubated fo... | Bioorg Med Chem 21: 5442-50 (2013) Article DOI: 10.1016/j.bmc.2013.06.002 BindingDB Entry DOI: 10.7270/Q2HD7X24 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| << First | Previous | Displayed 51 to 62 (of 62 total ) |