

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50336771 CHEMBL604158::sodium 1-amino-4-(4-fluorophenylamino)-9,10-dioxo-9,10-dihydroanthracene-2-sulfonate
SMILES: Nc1c(cc(Nc2ccc(F)cc2)c2C(=O)c3ccccc3C(=O)c12)S([O-])(=O)=O
InChI Key: InChIKey=HCTWKQCCDOTHIE-UHFFFAOYSA-M
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ecto-5'-nucleotidase (e5'NT) (Rattus norvegicus (Rat)) | BDBM50336771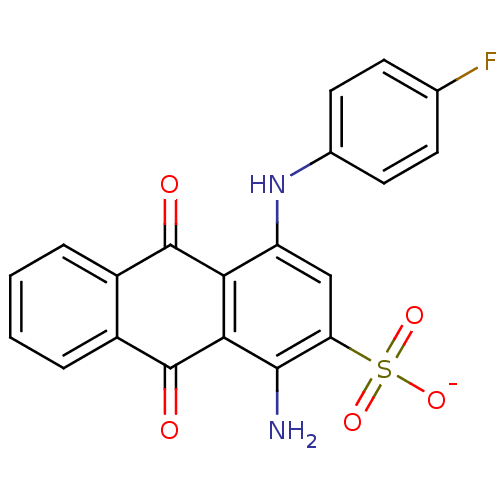 (CHEMBL604158 | sodium 1-amino-4-(4-fluorophenylami...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Inhibition of rat ecto-5'-nucleotidase expressed in Sf9 cells by capillary electrophoresis method | J Med Chem 53: 2076-86 (2010) Article DOI: 10.1021/jm901851t BindingDB Entry DOI: 10.7270/Q2DZ097V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 4 (RAT) | BDBM50336771 (CHEMBL604158 | sodium 1-amino-4-(4-fluorophenylami...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Antagonist activity against rat P2X4 receptor expressed in Xenopus laevis oocyte assessed as inhibition of alpha, beta-meATP-induced inward current b... | J Med Chem 54: 817-30 (2012) Article DOI: 10.1021/jm1012193 BindingDB Entry DOI: 10.7270/Q2VH5PTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y4 (Homo sapiens (Human)) | BDBM50336771 (CHEMBL604158 | sodium 1-amino-4-(4-fluorophenylami...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Antagonist activity at human P2Y4 receptor transfected in human 1321N1 cells assessed as inhibition of UTP-activated intracellular calcium mobilizati... | J Med Chem 60: 3020-3038 (2017) Article DOI: 10.1021/acs.jmedchem.7b00030 BindingDB Entry DOI: 10.7270/Q2G73H15 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic, P2X2 (RAT) | BDBM50336771 (CHEMBL604158 | sodium 1-amino-4-(4-fluorophenylami...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Antagonist activity against rat P2X2 receptor expressed in Xenopus laevis oocyte assessed as inhibition of alpha, beta-meATP-induced inward current b... | J Med Chem 54: 817-30 (2012) Article DOI: 10.1021/jm1012193 BindingDB Entry DOI: 10.7270/Q2VH5PTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||