

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CO[C@@H]1COCC[C@@H]1N[C@@H]1CC[C@@](CC(F)(F)F)(C1)C(=O)N1CCN(CC1)c1cc(ccn1)C(F)(F)F
InChI Key: InChIKey=WXRJQYAAIDCOMB-MOXWOTFGSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C-C chemokine receptor type 5 (Homo sapiens (Human)) | BDBM50337634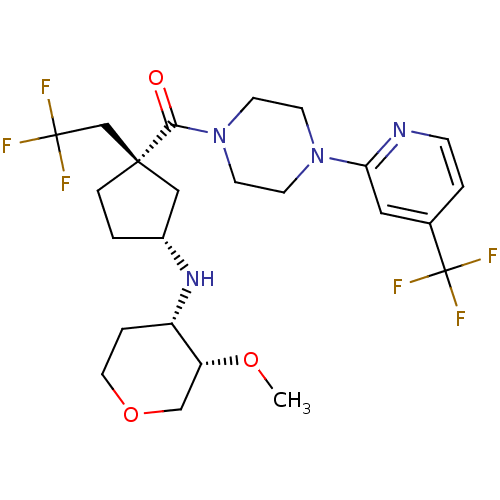 (((1S,3R)-3-((3S,4S)-3-methoxy-tetrahydro-2H-pyran-...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation Curated by ChEMBL | Assay Description Antagonist activity at human CCR5 receptor by chemotaxis assay | Bioorg Med Chem Lett 21: 1442-6 (2011) Article DOI: 10.1016/j.bmcl.2011.01.015 BindingDB Entry DOI: 10.7270/Q22J6C4C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| C-C chemokine receptor type 2 (Homo sapiens (Human)) | BDBM50337634 (((1S,3R)-3-((3S,4S)-3-methoxy-tetrahydro-2H-pyran-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation Curated by ChEMBL | Assay Description Displacement of [125I]MCP1 from human CCR2 after 30 mins by gamma counting | Bioorg Med Chem Lett 21: 1442-6 (2011) Article DOI: 10.1016/j.bmcl.2011.01.015 BindingDB Entry DOI: 10.7270/Q22J6C4C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| C-C chemokine receptor type 5 (Homo sapiens (Human)) | BDBM50337634 (((1S,3R)-3-((3S,4S)-3-methoxy-tetrahydro-2H-pyran-...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation Curated by ChEMBL | Assay Description Displacement of labeled MIP-1beta from human CCR5 receptor | Bioorg Med Chem Lett 21: 1442-6 (2011) Article DOI: 10.1016/j.bmcl.2011.01.015 BindingDB Entry DOI: 10.7270/Q22J6C4C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| C-C chemokine receptor type 2 (Homo sapiens (Human)) | BDBM50337634 (((1S,3R)-3-((3S,4S)-3-methoxy-tetrahydro-2H-pyran-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation Curated by ChEMBL | Assay Description Antagonist activity at human CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins | Bioorg Med Chem Lett 21: 1442-6 (2011) Article DOI: 10.1016/j.bmcl.2011.01.015 BindingDB Entry DOI: 10.7270/Q22J6C4C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| C-C chemokine receptor type 2 (Homo sapiens (Human)) | BDBM50337634 (((1S,3R)-3-((3S,4S)-3-methoxy-tetrahydro-2H-pyran-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation Curated by ChEMBL | Assay Description Displacement of MCP-Alexa 488 from CCR2 in human whole blood after 5 mins by flow cytometry | Bioorg Med Chem Lett 21: 1442-6 (2011) Article DOI: 10.1016/j.bmcl.2011.01.015 BindingDB Entry DOI: 10.7270/Q22J6C4C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||