

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50337736 CHEMBL1683455::N-((2S,4S)-4-(5-fluoropyrimidin-2-yl)-2-methyl-1-(4-(2-methyl-4-(trifluoromethyl)phenethyl)piperidin-1-ylsulfonyl)pentan-2-yl)-N-hydroxyformamide::US10322143, Compound 8h
SMILES: C[C@@H](C[C@@](C)(CS(=O)(=O)N1CCC(CCc2ccc(cc2C)C(F)(F)F)CC1)N(O)C=O)c1ncc(F)cn1
InChI Key: InChIKey=AYZCFZMCYDMFMY-DFBJGRDBSA-N
Data: 7 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Matrix metalloproteinase-14 (MMP14) (Homo sapiens (Human)) | BDBM50337736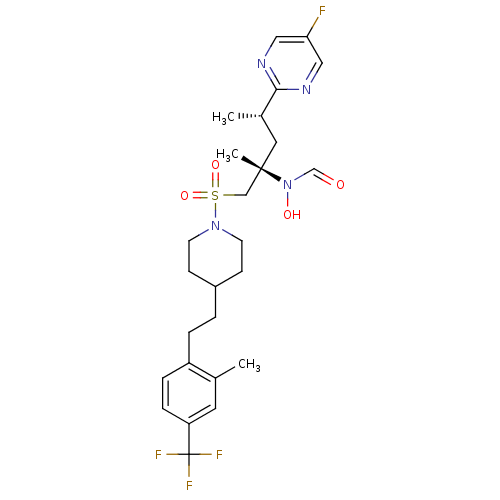 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of recombinant human MMP14 assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 4 (ADAMTS-4) (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of ADAMTS-4 assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of recombinant human MMP2 assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 4 (ADAMTS-4) (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca | Assay Description Protein concentrations were measured using the Micro BCA Protein Assay Kit (Pierce/Thermo Scientific, IL). Sample buffer was mixed with 5-25 μg ... | Bioorg Med Chem Lett 19: 4280-3 (2009) BindingDB Entry DOI: 10.7270/Q2BC41VN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of recombinant human MMP13 assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of recombinant human MMP1 assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Disintegrin and metalloproteinase domain-containing protein 17 (Homo sapiens (Human)) | BDBM50337736 (CHEMBL1683455 | N-((2S,4S)-4-(5-fluoropyrimidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of TACE assessed as substrate cleavage by measuring increase in fluorescence after 16 hrs | Bioorg Med Chem Lett 21: 1376-81 (2011) Article DOI: 10.1016/j.bmcl.2011.01.036 BindingDB Entry DOI: 10.7270/Q2DV1K5M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||