

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50338363 CHEMBL1682893::rac-6-[1-Hydroxy-1-(1H-imidazol-4-yl)-2-methylpropyl]-Nmethyl-2-naphthamide
SMILES: CNC(=O)c1ccc2cc(ccc2c1)C(O)(C(C)C)c1cnc[nH]1
InChI Key: InChIKey=XEYZCKZMDNQROP-UHFFFAOYSA-N
Data: 6 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 17A1 (Rattus norvegicus (Rat)) | BDBM50338363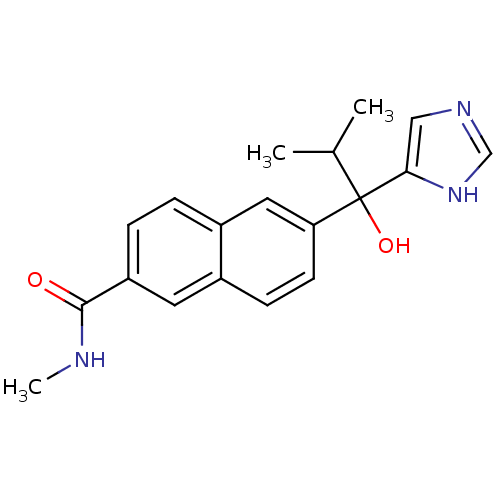 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Inhibition of 17,20-lyase activity of Sprague-Dawley rat testicular microsomal CYP17A1 using [1,2-3H]-17a-hydroxyprogesterone as substrate after 15 m... | Bioorg Med Chem 19: 6383-99 (2011) Article DOI: 10.1016/j.bmc.2011.08.066 BindingDB Entry DOI: 10.7270/Q2X63NDF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 17A1 (Homo sapiens (Human)) | BDBM50338363 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Inhibition of 17,20-lyase activity of human CYP17A1 | Bioorg Med Chem 19: 6383-99 (2011) Article DOI: 10.1016/j.bmc.2011.08.066 BindingDB Entry DOI: 10.7270/Q2X63NDF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50338363 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd Curated by ChEMBL | Assay Description Inhibition of human CYP3A4 | Bioorg Med Chem 19: 1751-70 (2011) Article DOI: 10.1016/j.bmc.2011.01.017 BindingDB Entry DOI: 10.7270/Q2X92C9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 17A1 (Rattus norvegicus (Rat)) | BDBM50338363 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd Curated by ChEMBL | Assay Description Inhibition of 17,20 lyase activity in Sprague-Dawley rat testicular microsomes | Bioorg Med Chem 19: 1751-70 (2011) Article DOI: 10.1016/j.bmc.2011.01.017 BindingDB Entry DOI: 10.7270/Q2X92C9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 17A1 (Homo sapiens (Human)) | BDBM50338363 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd Curated by ChEMBL | Assay Description Inhibition of 17,20 lyase activity in human | Bioorg Med Chem 19: 1751-70 (2011) Article DOI: 10.1016/j.bmc.2011.01.017 BindingDB Entry DOI: 10.7270/Q2X92C9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50338363 (CHEMBL1682893 | rac-6-[1-Hydroxy-1-(1H-imidazol-4-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Inhibition of recombinant human CYP3A4 assessed as conversion of testosterone into 6-hydroxytestosterone after 30 mins by HPLC analysis | Bioorg Med Chem 19: 6383-99 (2011) Article DOI: 10.1016/j.bmc.2011.08.066 BindingDB Entry DOI: 10.7270/Q2X63NDF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||