

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50355892 CHEMBL1910035
SMILES: Cc1ccc(cc1)[C@H](N1CCN(CC1)C(=O)NC1CCCCC1)c1ccc(Cl)cc1Cl
InChI Key: InChIKey=LRFGNVCBHZJPNK-DEOSSOPVSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50355892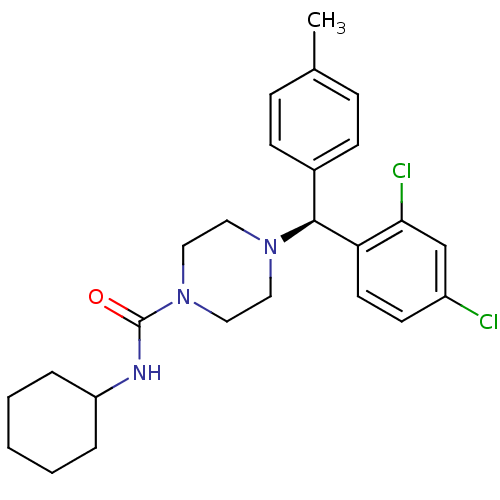 (CHEMBL1910035) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Materia Medica Curated by ChEMBL | Assay Description Displacement of [3H]-CP-55940 from human CB1 receptor expressed in CHO cells pretreated for 10 mins measured after 3 hrs by scintillation counting | Eur J Med Chem 46: 5310-6 (2011) Article DOI: 10.1016/j.ejmech.2011.08.030 BindingDB Entry DOI: 10.7270/Q22J6C85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM50355892 (CHEMBL1910035) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 227 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Materia Medica Curated by ChEMBL | Assay Description Displacement of [3H]-CP-55940 from human CB2 receptor expressed in CHO cells pretreated for 10 mins measured after 3 hrs by scintillation counting | Eur J Med Chem 46: 5310-6 (2011) Article DOI: 10.1016/j.ejmech.2011.08.030 BindingDB Entry DOI: 10.7270/Q22J6C85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50355892 (CHEMBL1910035) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Materia Medica Curated by ChEMBL | Assay Description Antagonist activity at human CB1 receptor expressed in CHO cells coexpressing Galpha15/16 assessed as inhibition of CP55940-induced Ca2+ release afte... | Eur J Med Chem 46: 5310-6 (2011) Article DOI: 10.1016/j.ejmech.2011.08.030 BindingDB Entry DOI: 10.7270/Q22J6C85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||