

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50380204 CHEMBL2011405::US10406136, Compound 6
SMILES: [#6]-[#8]-c1c(-[#8])cc(cc1-[#8])-[#6]-1-[#6]-[#6](=O)-c2c(-[#8])c(-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](\[#6])-[#6])c(-[#8])cc2-[#8]-1
InChI Key: InChIKey=GMGRYHOHJJROMP-OVCLIPMQSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Butyrylcholinesterase (BChE) (Equus caballus (Horse)) | BDBM50380204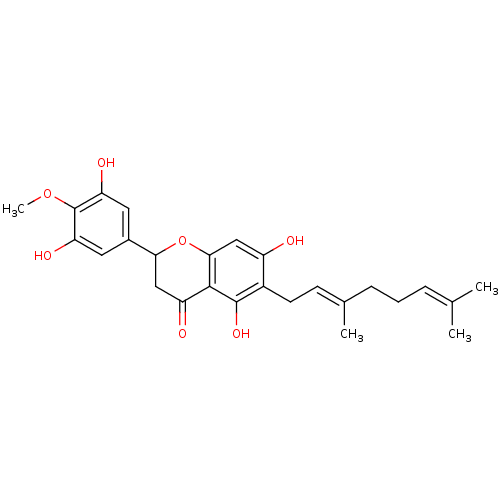 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Mixed type inhibition of equine BChE using butyrylthiocholine iodide as substrate by Lineweaver-Burk double-reciprocal-plot and dixon plot analysis | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50380204 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.01E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Mixed type inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate by Lineweaver-Burk double-reciprocal-plot and dixon plot ... | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50380204 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INDUSTRY-ACADEMIC COOPERATION FOUNDATION GYEONGSANG NATIONAL UNIVERSITY US Patent | Assay Description Specifically, in order to determine IC50 of the compounds for neuraminidase, 0.01 U/ml of neuraminidase (EC. 3.2.1.8, C. perfringens, SIGMA, N2876) a... | US Patent US10406136 (2019) BindingDB Entry DOI: 10.7270/Q2NS0X86 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50380204 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
INDUSTRY-ACADEMIC COOPERATION FOUNDATION GYEONGSANG NATIONAL UNIVERSITY US Patent | Assay Description Specifically, in order to determine IC50 of the compounds for neuraminidase, 0.01 U/ml of neuraminidase (EC. 3.2.1.8, C. perfringens, SIGMA, N2876) a... | US Patent US10406136 (2019) BindingDB Entry DOI: 10.7270/Q2NS0X86 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50380204 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE assessed as acetylthiocholine iodide hydrolysis after 10 mins preincubation by spectrophotometry | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Butyrylcholinesterase (BChE) (Equus caballus (Horse)) | BDBM50380204 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of equine BChE assessed as butyrylthiocholine iodide hydrolysis after 10 mins preincubation by spectrophotometry | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||