 Found 15 hits for monomerid = 50385915
Found 15 hits for monomerid = 50385915 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine/threonine-protein kinase Nek2
(Homo sapiens (Human)) | BDBM50385915
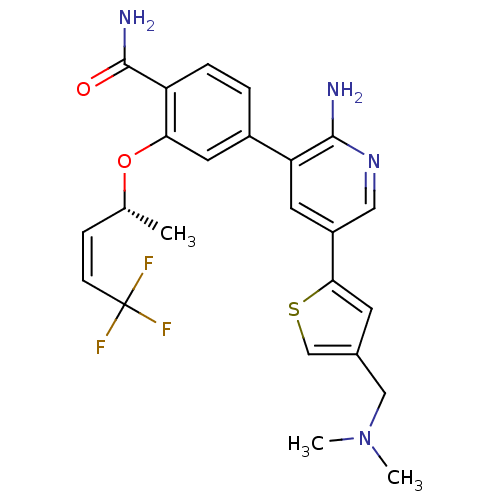
(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Nek2 using 5-FAM-KKLNRTLSVA-COOH as substrate after 1 hr by caliper method |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.57E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of CDK2 by caliper method |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.11E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of ABL at 1 uM |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Fyn
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 750 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of FYN incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lyn
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.36E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of LYN incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase CHK2
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of CHK2 incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of MET incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 840 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of SRC incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of GSK3beta incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Nek2
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Nek2-mediated C-Nap1 phosphorylation in human U2OS cells after 3 hrs by immunofluorescence micrscopy |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase PLK1
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.82E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Plk1 using 5-FAMRRRAGALMDASFEEQ- CONH2 as substrate after 75 mins by caliper method |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Dual specificity protein kinase TTK
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of MPS1 by caliper method |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.94E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A kinase by caliper method |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase D2
(Homo sapiens (Human)) | BDBM50385915

(CHEMBL2042135)Show SMILES C[C@@H](Oc1cc(ccc1C(N)=O)-c1cc(cnc1N)-c1cc(CN(C)C)cs1)\C=C/C(F)(F)F |r| Show InChI InChI=1S/C24H25F3N4O2S/c1-14(6-7-24(25,26)27)33-20-10-16(4-5-18(20)23(29)32)19-9-17(11-30-22(19)28)21-8-15(13-34-21)12-31(2)3/h4-11,13-14H,12H2,1-3H3,(H2,28,30)(H2,29,32)/b7-6-/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
The Institute of Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of PKD2 incubated for 30 mins prior to substrate addition study done at apparent ATP Km for enzyme |
J Med Chem 55: 3228-41 (2012)
Article DOI: 10.1021/jm201683b
BindingDB Entry DOI: 10.7270/Q2Q81F4F |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data